|
Article Information
|
Authors:
Arthur M. Spickett1
I. Heloise Heyne1
Roy Williams2
Affiliations:
1Parasites, Vectors and Vector-borne Diseases Programme, ARC-Onderstepoort Veterinary Institute,
South Africa
2Veterinary Geographic Information Systems Programme, ARC-Onderstepoort Veterinary Institute,
South Africa
Correspondence to:
Arthur Spickett
Email:
SpickettAM@arc.agric.za
Postal address:
Private Bag X05, Onderstepoort 0110,
South Africa
Dates:
Received: 12 Nov. 2010
Accepted: 12 Jan. 2011
Published: 14 Sept. 2011
Republished: 09 Nov. 2011
How to cite this article:
Spickett, A.M., Heyne, I.H. & Williams, R., 2011, ‘Survey of the livestock ticks of the North West province, South Africa’,
Onderstepoort Journal of Veterinary Research 78(1), Art. #305, 12 pages.
doi:10.4102/ojvr.v78i1.305
Note:
This article was republished with the corrected name of Ingard H. Heyne.
Copyright Notice:
© 2011. The Authors. Licensee: AOSIS OpenJournals. This work
is licensed under the Creative Commons Attribution License.
ISSN:
0030-2465 (print)
ISSN:
2219-0635 (online)
|
|
|
|
Survey of the livestock ticks of the North West province, South Africa
|
|
In This Original Research...
|
Open Access
|
• Abstract
• Introduction
• Materials and methods
• Study locality
• Survey design and execution
• Results and discussion
• Number of ticks, species composition and collections
• Tick species distribution and seasonality
• Amblyomma hebraeum
• Hyalomma rufipes
• Hyalomma truncatum
• Rhipicephalus appendiculatus
• Rhipicephalus (Boophilus) decoloratus
• Rhipicephalus (Boophilus) microplus
• Rhipicephalus evertsi evertsi
• Rhipicephalus evertsi mimeticus
• Rhipicephalus simus
• Tick-borne disease serology
• Conclusion
• Acknowledgements
• References
|
|
Ticks, as vectors of disease and damage agents, impact directly and indirectly on the economy of the livestock industry in southern Africa. This study
surveyed the occurrence and distribution of ticks infesting livestock across the North West province, South Africa. During three phases in consecutive
years, officers of the provincial Veterinary Department collected specimens monthly from livestock hosts at specified sites across the province. Data
analysis constituted the fourth phase of the study. A total of 1090 collections from 265 sites yielded 42 566 tick specimens, comprising 22 different
tick species (18 ixodids, 4 argasids). The specimens represent all of the major tick vectors of disease that occur in South Africa. The major tick-borne
diseases (i.e. heartwater, both African and Asiatic bovine babesiosis and anaplasmosis) were found to be prevalent mainly in the north-eastern region of
the province, which also displayed the highest tick species diversity. The central region appears transitory to some of the major vectors. Although some
tick species were contained within specific regions, others were widespread across the province. Associated serology data show that most herds sampled in
areas endemic for babesiosis and anaplasmosis in the north-eastern region are endemically unstable and at risk to these tick-borne diseases should vector
control measures become ineffective.
Ixodid ticks are economically of the most important external parasites of livestock in the tropical and sub-tropical parts of the world, including South Africa.
Heavy infestation can cause loss of blood, reduce live weight gain rates and lower milk yield, whilst the long-mouthed ticks downgrade the quality of the hides
and cause secondary infections (De Castro 1997). They are also recognised worldwide as major vectors of a number of disease-causing pathogens in humans and
domestic and game animals, including arboviruses, rickettsiae, spirochaetes and parasitic protozoa. They act as reservoirs and/or multipliers of these organisms
and transmit a number of important tick-borne diseases such as heartwater, bovine babesiosis, anaplasmosis and theileriosis. Some ixodid ticks also produce toxins
that cause paralysis in sheep (Spickett & Heyne 1988) and sweating sickness in calves (Jongejan & Uilenberg 1994).
Ticks therefore impact both directly (by virtue of heavy infestations) and indirectly (through the transmission of tick-borne diseases) at an economical level as
well as in a social context. At the macro-economic level, exports and the commercial production of protein may be affected and at the micro-economic level the
subsistence economy of the resource-poor farmer may be at risk. Whilst little actual data exist, it was estimated already some 30 years ago that mortality losses
attributed to tick-borne diseases, together with acaricide and vaccine costs, were in the region of R70 million per annum (Van Rensburg 1981), whilst a panel of
the Food and Agriculture Organization placed total losses in South Africa at R550 million per annum in 1985. However, these are estimates at best and many resources
would have to be harnessed in order to determine more accurate figures regarding the economic impact of ticks. Only one study – on heartwater, which is
probably the most important tick-borne disease in South Africa – has been attempted (Minjauw 2005), with total annual losses estimated at R189.6 million.
Paramount to assessing the impact of vectors and disease agents are the identification of the species involved, their distribution and their association with
disease. Long-term research since the time of Arnold Theiler in the early 20th century, including the last comprehensive tick survey in South Africa by Gertrud
Theiler in 1957 (Theiler 1962), has placed South Africa in the fortunate position of having a wide knowledge of ticks and their associated disease epidemiologies.
These findings are documented in wide-ranging publications, with the most important disease-oriented knowledge being condensed into two publications (Coetzer &
Tustin 2004; Walker, Keirans & Horak 2000). In the South African context, some 90 ixodid and 25 argasid tick species have been identified. Of these, 35 are
normally associated with domestic animals and approximately 15 are considered of economic importance. They transmit, or are associated with, some 23 diseases or
health syndromes (Norval & Horak 2004).
In the context of ticks and tick-borne diseases affecting livestock resources, the Directorate of Veterinary Services in the North West province (Department of
Agriculture), commissioned a survey to determine tick species presence, their seasonal occurrence, contribution to livestock diseases and distribution across the
province.
Study locality
The survey was conducted over a total area of 116 320 km² in the North West province of South Africa (between approximately 24°15’S,
22°30’E and 28°15’S, 28°30’E). The area may, according to Acocks (1988), be sectored roughly into three main regions based
on vegetation (Figure 1). The north-eastern region consists of bushveld (Mixed, Sour and Sourish-mixed Bushveld, the latter interspersed with Bankenveld and
Turf Thornveld). The central region has an equally mixed vegetation, consisting of Dry and Sandy Cymbopogon–Themeda veld with Sourish-mixed
Bushveld in its northern reaches. The western, more arid region of the province consists mainly of Kalahari Thornveld.
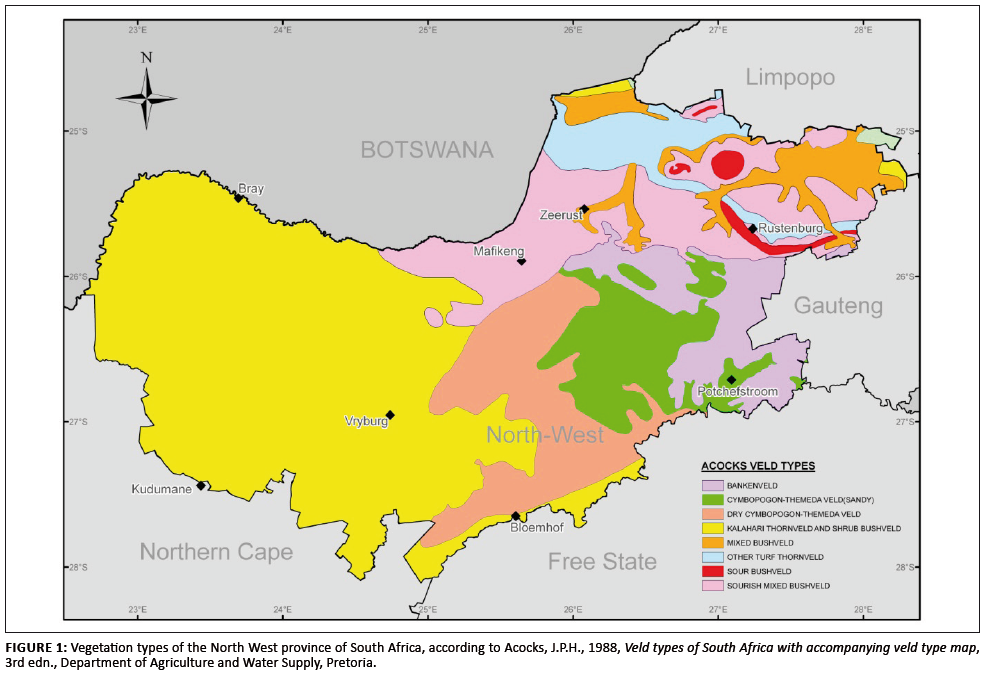 |
FIGURE 1: Vegetation types of the North West province of South Africa,
according to Acocks, J.P.H., 1988, Veld types of South Africa with accompanying veld type map,
3rd edn., Department of Agriculture and Water Supply, Pretoria.
|
|
The climate of the province is characterised by well-defined seasons: hot summers and cool, sunny winters. The climate and rainfall vary from the more mountainous
and wetter eastern region to the drier, semi-desert plains of the Kalahari in the west. The rainy season usually falls between October and March. Rainfall is
highly variable, with regard to both time and region. On average, the western part of the province receives less than 300 mm rainfall per annum, the central
part around 550 mm and the eastern and south-eastern parts receive more than 600 mm.
There are wide seasonal and daily variations in temperature. Summers are very hot (daily average maximum temperatures of 32 ºC in January) and winters are
mild to cold (average daily minimum in July is 1 ºC). The far western part is arid, encompassing the eastern portion of the Kalahari Desert. The central part
of the province is typically semi-arid, whilst the eastern region is predominantly temperate.
Agriculture is the second most important economic activity. The eastern, wetter region of the province largely supports livestock and crop farming, whilst the
central and southern regions are dominated by wheat and maize farming. Livestock and game farming occur in the semi-arid western region of the province. Tourism
is considered to have a major growth potential as the province has a rich natural heritage and a network of well-managed, malaria-free national and provincial
parks and nature reserves, which boast the ’Big Five’. According to the Department of Agriculture, Forestry and Fisheries (2004) livestock production
revolves around the estimated 1.77 million cattle, 729 000 sheep and 753 000 goats in the province. Approximately 43% of cattle, 46% of sheep and 47% of
goats are owned by the communal sector. The majority of cattle are found in the north-eastern region (42%), with 34% and 24% being in the central and western
regions, respectively. Sheep and goats are farmed predominantly in the western region (44% and 46%, respectively), whilst only to a limited extent in the
north-eastern and central regions (sheep: 29% and 27%, respectively; goats: 34% and 20%, respectively). It is this considerable livestock resource that could be
adversely affected by ticks and tick-borne diseases.
Survey design and execution
The survey was conducted in four phases over a number of years since 2001. The first three phases included monthly tick collections from identified sites in the
north-eastern, central and western regions during three consecutive years (Figure 2).
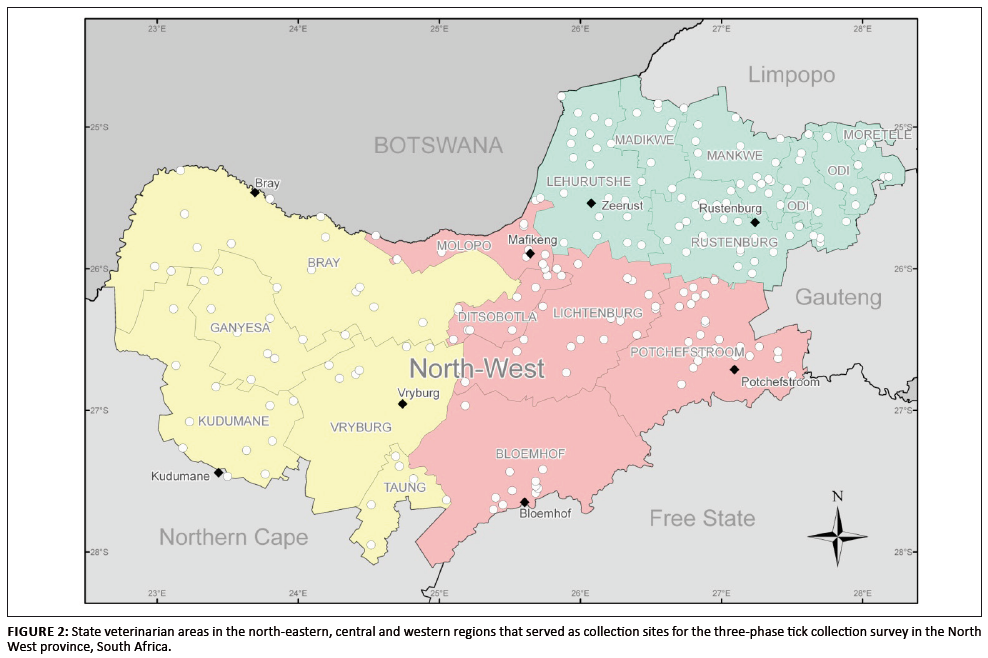 |
FIGURE 2: State veterinarian areas in the north-eastern,
central and western regions that served as collection sites for the three-phase tick collection survey in the North
West province, South Africa.
|
|
Collection sites were selected from 1:250 000 charts and subsequently between four and 41 collection sites, depending on spatial distribution, logistics and
collectors’ time constraints, were assigned to specific areas in each of the three regions (Table 1). Areas considered marginal for specific tick species
were included in the selected sites. A total of 114 sites were selected in the north-eastern region (collection phase 1), 116 in the central region (collection
phase 2) and 61 in the western region (collection phase 3). As comprehensive coverage as possible of the province was sought to allow for tick distribution mapping.
Collection sites were not assigned to the immediate north-east and north-west of Bloemhof (central region) owing to crop farming predominating in these areas.
|
TABLE 1: Sampling areas in three regions of the North West province, South Africa.
|
During each collection phase, nominated animal health technicians from each region received in-house training in basic tick identification, collection and specimen
handling before collection commenced at their specified sites. The fourth phase (performed at the Onderstepoort Veterinary Institute [OVI]) involved species
identification, database acquisition, distribution mapping using ArcGIS (ESRI) software and reporting.
Ticks were collected off one side of livestock hosts – mainly cattle, goats and sheep – using forceps. Each collection was placed in 70% alcohol in a
specimen bottle for dispatch to the OVI for identification and recording. Only one collector, in the Bray area, consistently sampled chicken coops. Owing to prior
training collectors were able to perform basic tick species identification in situ, so that as wide a range of species present on the animal could be collected.
To gain data on species seasonality, monthly collections from most sites were attempted. Tick collections were, however, disrupted to some extent during the second
phase (central region) because of a swine fever outbreak in the Eastern Cape, which required secondment of personnel to assist with containment operations.
In some areas of the north-eastern region, a high incidence of both Rhipicephalus (Boophilus) decoloratus and Rhipicephalus (Boophilus) microplus,
the vectors of bovine babesiosis, was evident. A standard immunofluorescent antibody test (Joyner et al. 1972) and a competitive inhibition
enzyme-linked immunosorbent assay (Ndung’u et al. 1995; Visser et al. 1992) were used to test blood samples for antibodies to Babesia bovis
and Babesia bigemina, and Anaplasma marginale, respectively. Sera collected from 13 properties in the affected areas (from at least
30 cattle per herd, 9–12 months of age) were tested to determine the antibody status of these herds to African and Asiatic babesiosis and anaplasmosis.
Number of ticks, species composition and collections
A total of 42 566 ticks (from 1190 collections across 265 collection sites) were received for identification. The breakdown of collections is as follows:
(1) north-eastern region: 21 045 specimens from 429 collections
(2) central region: 10 391 specimens from 398 collections
(3) western region: 11 130 from 363 collections.
Owing to the collection method employed almost all the ticks received were adults (97.9%), with roughly equal numbers of males and females. The immatures
(nymphs = 1.8%; larvae = 0.3%), mostly Rhipicephalus evertsi evertsi and four nymphs of Otobius megnini, were collected more by chance than intention
from the ears of cattle.
Altogether 22 tick species were identified (18 from six Ixodidae genera; four from three Argasidae genera). The main tick species were all hard ticks (Ixodidae) and
included R. evertsi evertsi (31.5%), Hyalomma rufipes (22.4%), Amblyomma hebraeum (17.3%), R. (B.) decoloratus (12.5%), Rhipicephalus
appendiculatus (11.2%), Hyalomma truncatum (2.3%), R. (B.) microplus (1.6%), Rhipicephalus evertsi mimeticus (0.5%) and
Rhipicephalus simus (0.21%). Other ticks of lesser importance or those collected in low numbers or during only a few collections, as well as the Argasidae
collections, are as shown in Table 2.
|
TABLE 2: Ixodid ticks of lesser importance and argasid ticks identified from each of the three collection regions.
|
Monthly collections over a period of 10–12 months were received from 70 collection sites, which allowed the seasonal occurrence of the major tick vectors to
be determined. Monthly collections from a further 175 collection sites were received over a period varying from 1 to 9 months.
Tick species distribution and seasonality
The distribution patterns and seasonality of the major tick species collected are described below.
Amblyomma hebraeum
According to Norval and Horak (2004), Amblyomma hebraeum is present in grassed bushveld and wooded savanna regions. In the present survey,
A. hebraeum accounted for 17.3% of the collected ticks and appears well established in the Mixed and Sourish-mixed Bushveld areas of
the North West province (Figure 3). It is present mainly in the north-eastern and northern portion (Mixed Bushveld) of the central region, with
singular intrusions into the Kalahari Thornveld south-east of Vryburg and west of Mafikeng. The latter distribution appears to be established
populations because they were consistently collected during the summer months at these sites.
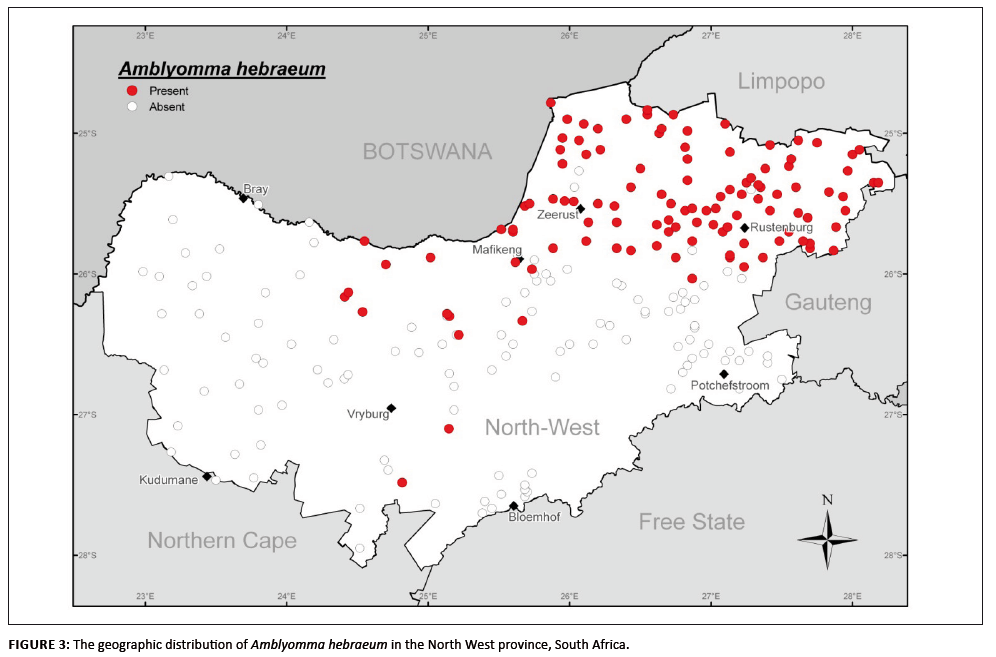 |
FIGURE 3: The geographic distribution of Amblyomma hebraeum in the North West province, South Africa.
|
|
Adult ticks prefer large hosts such as cattle and large wild ruminants, but also infest sheep and goats (Horak et al. 1987; Walker et al. 2003).
In this survey 90.8% of A. hebraeum specimens were recovered from cattle, 4.9% from goats and 3.3% from sheep (1.0% were not stipulated). The adults of
this species were present throughout the year, with a high incidence in both the north-eastern and central regions during November and December (summer),
similar to observations by Rechav (1982) for the Eastern Cape. Higher numbers of adults were found in the central region between March and May (autumn) than
in the north-eastern region during these months.
Hyalomma rufipes
H. rufipes (previously the subspecies Hyalomma marginatum rufipes [Apanaskevich & Horak 2008]) is a vector of the Crimean–Congo
haemorrhagic fever virus. It is the most widely distributed species of this tick genus in South Africa, occurring in the greater part of the country
(Norval & Horak 2004). It was found distributed widely over the whole of the North West province, with it being absent from only eight of the collection
sites (Figure 4). The life cycle may be completed in one year and peak numbers of adults were evident in the summer months (November–February).
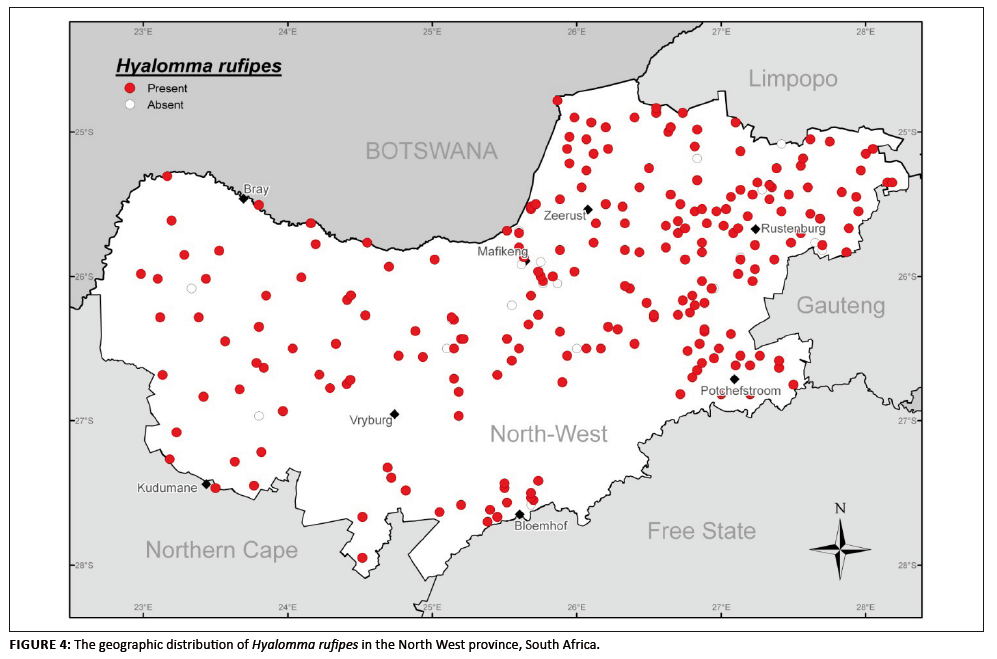 |
FIGURE 4: The geographic distribution of Hyalomma rufipes in the North West province, South Africa.
|
|
H. rufipes accounted for 22.4% of the total number of specimens collected in this survey. Of these, 97.2% were from cattle, 1.5% from sheep and 0.5% were
found on goats (0.8% were not assigned a host).
Hyalomma truncatum
H. truncatum is widely distributed over the whole of the North West province (Figure 5) and was present in all the ecological zones sampled. Of the
adult H. truncatum specimens collected, 89.9% were from cattle, 0.8% from sheep and 8.5% from goats; 0.8% were not assigned a host. H. truncatum
accounted for only 2.3% of the total number of specimens collected. Some strains of this species transmit a toxin that causes an epitheliotrophic condition
called sweating sickness, which affects young calves in particular (Neitz 1959). The tendency for adults to attach preferentially in the tail switch of cattle
(where it is easily overlooked during sampling) may explain why this tick was not collected at more of the collection sites. Its seasonal occurrence in the North
West province confirms its annual life cycle, with adults reaching peak numbers between January and March.
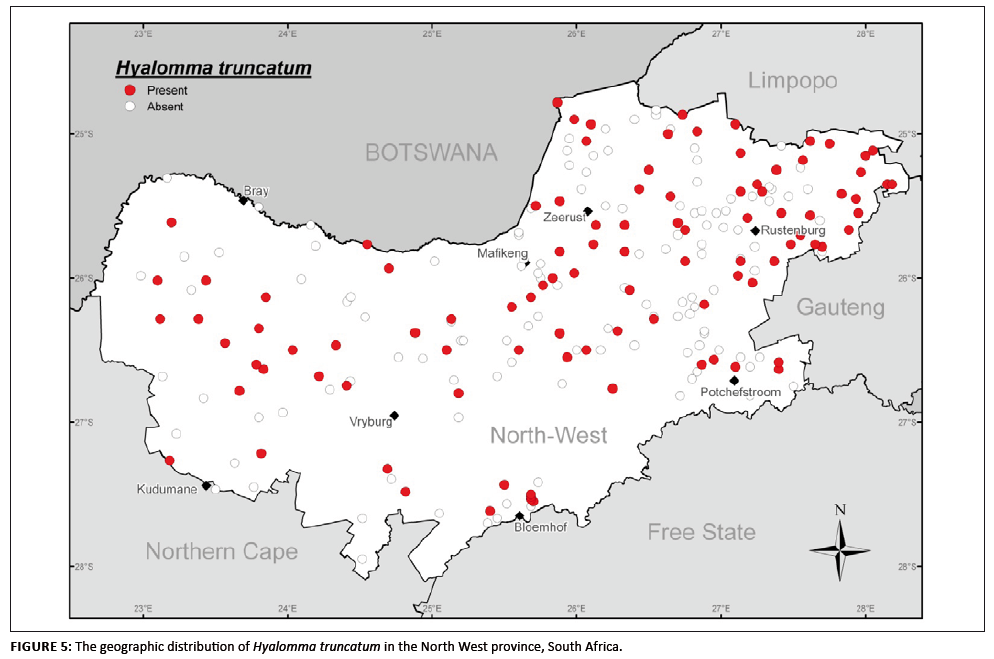 |
FIGURE 5: The geographic distribution of Hyalomma truncatum in the North West province, South Africa.
|
|
Rhipicephalus appendiculatus
R. appendiculatus is restricted to the higher-rainfall, eastern regions of South Africa (Walker et al. 2000) and according to Theiler (1962) does
not occur in open grassland without bush; it prefers tall grass interspersed with trees. Accordingly, this species was found well established in the north-eastern
region of the province and in the bushveld areas of the northern reaches of the central region around Mafikeng (Figure 6). The remainder of the province, especially
the western region, appears to be ecologically unsuited to the establishment of this tick species.
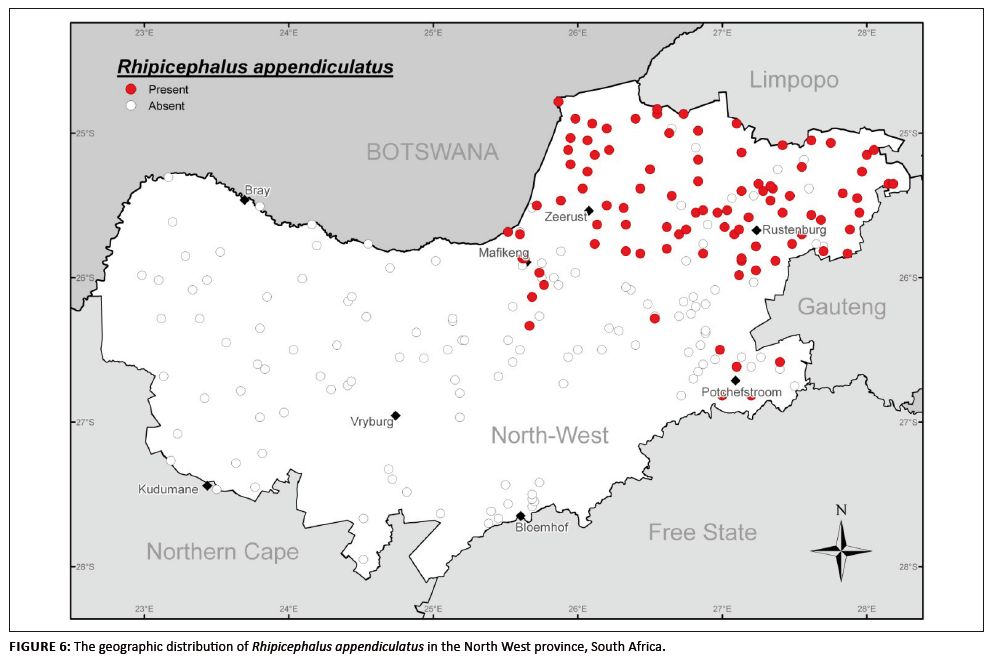 |
FIGURE 6: The geographic distribution of Rhipicephalus appendiculatus in the North West province, South Africa.
|
|
This species accounted for 11.2% of the specimens collected. Of the total number of adults collected, 86.9% were from cattle, 7.4% from goats and 5.0% from sheep;
the remaining 0.7% were not assigned a host. Adult activity commenced only during the summer months (between December and March), as reported for the Eastern Cape
by Rechav (1982).
Rhipicephalus (Boophilus) decoloratus
The ticks we have chosen to name R. (B.) decoloratus and R. (B.) microplus were originally known as Boophilus decoloratus and Boophilus
microplus, respectively. However, based on molecular evidence (Beati & Keirans 2001; Murrell, Campbell & Barker 2000), they were included in a 2002
world list of valid tick names as R. (B.) decoloratus and R. (B.) microplus (Horak, Camicas & Keirans 2002). Guglielmone et al.
(2010) have subsequently omitted the subgenus and these ticks appear in their list of valid tick names as R. decoloratus and R. microplus. We chose to
include the subgenus in our nomenclature.
R. (B.) decoloratus represented 12.5% of the collections in this survey, with 98.1% of specimens being collected from cattle, only 0.5% from sheep and 0.3%
from goats. The remaining 1.1% were not assigned hosts. This distribution confirms its host preference for large ungulates (Mason & Norval 1980). The species
normally occurs in temperate savanna regions, typically in grass- and woodland areas used by cattle, and tends to be absent in drier areas (Walker et al. 2003).
It is well established in the grassed Bushveld biomes (especially the Sourish-mixed Bushveld) of the north-eastern region and northern reaches of the central region of
the North West province, around Rustenburg, Zeerust, Mafikeng and Potchefstroom (Figure 7). The tick also inhabits seemingly suitable habitats (probably created by
irrigated land use) in the Cymbopogon–Themeda veld near Bloemhof and the Kalahari Thornveld near Kudumane and Vryburg in the western region.
Apparently, suitably high temperatures – well above the developmental threshold of 10 ºC (Spickett & Heyne 1990) – and host availability yielded
high numbers of this one-host species, even during the winter months. Peak numbers were evident in December and January and again from March to July. Since R. (B.)
decoloratus is a vector of B. bigemina, African babesiosis may therefore be a threat throughout the year.
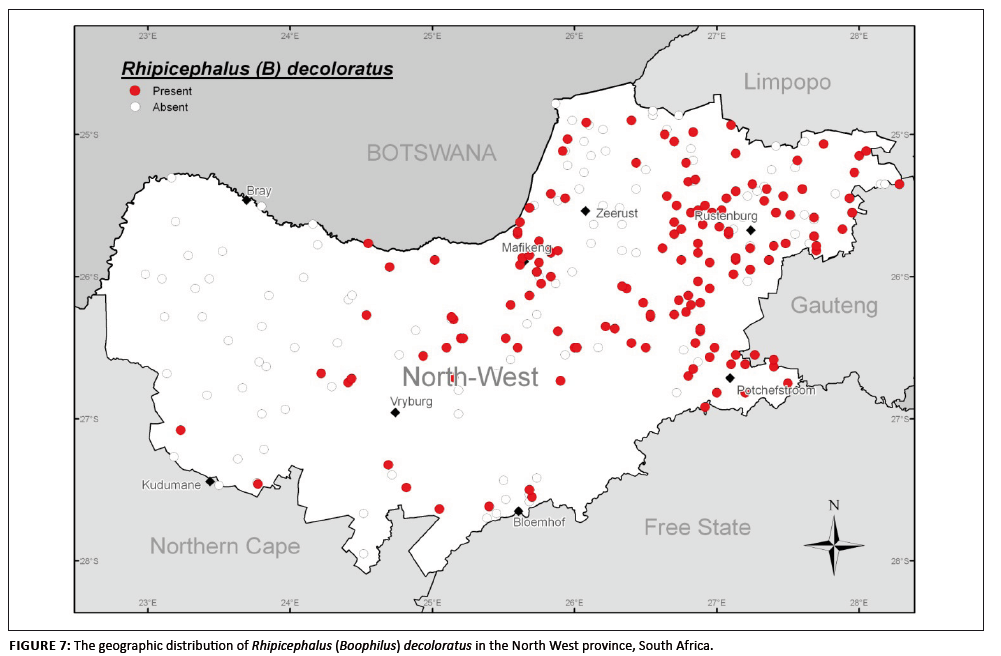 |
FIGURE 7: The geographic distribution of Rhipicephalus (Boophilus) decoloratus in the North West province, South Africa.
|
|
Rhipicephalus (Boophilus) microplus
R. (B.) microplus, as R. (B.) decoloratus, inhabits savanna climatic regions in wooded grasslands used as cattle pasture (Walker et al. 2003).
R. (B.) microplus (Figure 8) was found to have a more limited distribution compared to that of R. (B.) decoloratus, being confined
to the Sourish-mixed Bushveld of the north-eastern region (around Rustenburg, north-east of Potchefstroom and north of Zeerust). The species was also found in an
isolated pocket near Bloemhof, probably introduced by cattle and finding suitable habitats here.
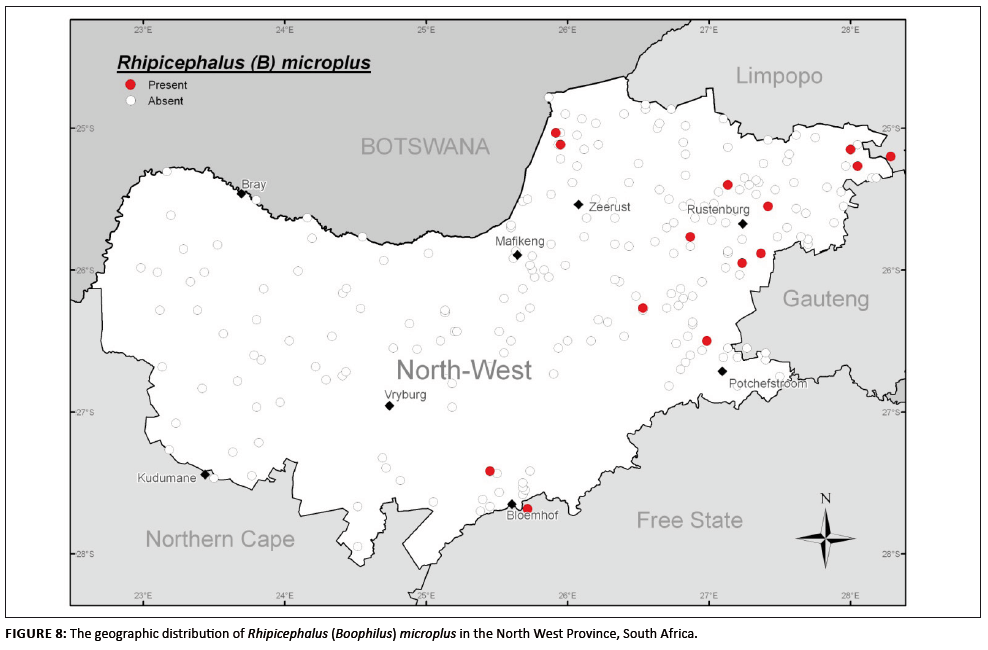 |
FIGURE 8: The geographic distribution of Rhipicephalus (Boophilus) microplus in the North West Province, South Africa.
|
|
Cattle are considered the only effective hosts of R. (B.) microplus (Mason & Norval 1980), to the extent that this species is absent in game
parks, where no cattle occur (Horak et al. 1986). Recently, goats have been implicated as alternate hosts for this species, but in the presence of cattle
in order to maintain populations (Nyangiwe & Horak 2007). In the present survey 81.8% of R. (B.) microplus specimens were collected from cattle and,
significantly, a relatively high 7.7% were found on goats. None were found on sheep. The remaining 10.5% of the collected R. (B.) microplus specimens were
not assigned a host by the collectors, but were most probably collected off cattle.
The seasonality of R. (B.) microplus was similar to that of R. (B.) decoloratus, with peak numbers occurring during the autumn months
of April and May.
Rhipicephalus evertsi evertsi
R. evertsi evertsi is widely distributed and common on livestock throughout much of Africa, occurring in desert, steppe, savanna and temperate climatic
regions (Walker et al. 2003). It has the most widespread distribution of species in the genus Rhipicephalus in Africa and has an extensive host
range (Walker et al. 2000). It was the most commonly collected species in the North West province (31.5% of all specimens), being distributed over the whole
province and absent at only two collection sites (Figure 9). In this survey, the adults that were collected came from cattle (79.1%), goats (15.9%) and sheep (3.9%),
whilst the remaining 1.1% were unaccounted for as to host. Peak activity was recorded early in autumn (May), although high numbers were also recorded during the summer
months.
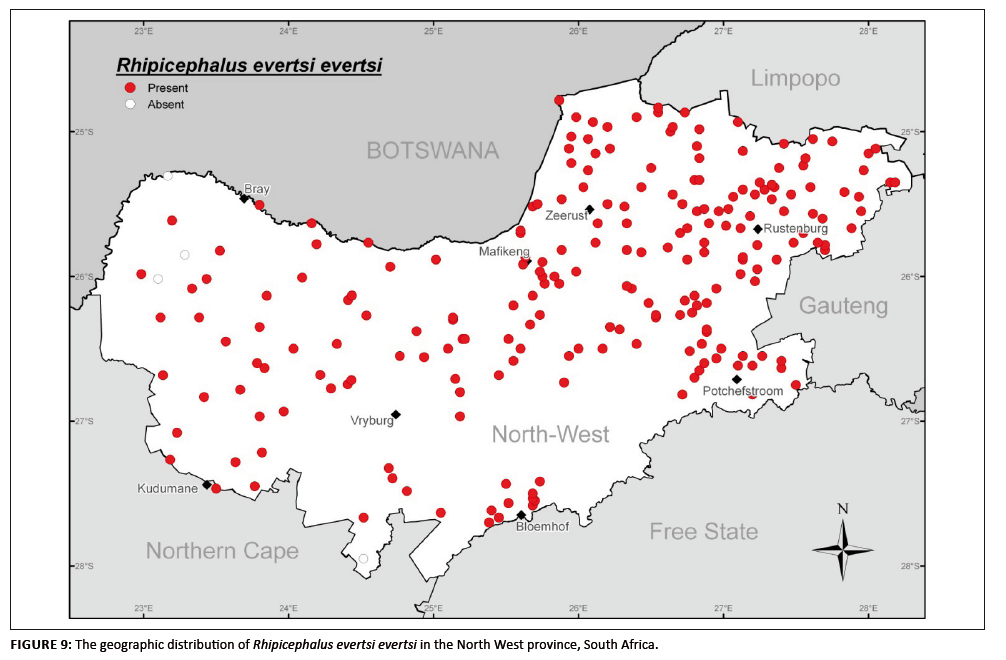 |
FIGURE 9: The geographic distribution of Rhipicephalus evertsi evertsi in the North West province, South Africa.
|
|
Rhipicephalus evertsi mimeticus
Although of minimal economic importance, this species is interesting in that it is recorded as occurring in the arid regions of Angola, Namibia and Botswana
(Theiler 1962). With regard to morphology and biology it is very similar to its subspecies R. evertsi evertsi and can also cause paralysis in sheep, mainly
lambs, owing to secreting a toxin in the saliva whilst feeding (Gothe 1999).
This species has infiltrated the North West province, with suitable habitat conditions enabling it to establish populations in the western region (Figure 10),
possibly by means of sheep introduced from Botswana or Namibia, and thus likely displacing R. evertsi evertsi at these localities. Recurring monthly
collections of relatively high numbers off sheep from these sites confirmed the establishment of viable R. evertsi mimeticus populations. Being subspecies,
R. evertsi mimeticus and R. evertsi evertsi should interbreed, yet both were found to be morphologically distinct and could easily be distinguished
taxonomically at the same localities, which may indicate some degree of speciation. This observation needs further investigation.
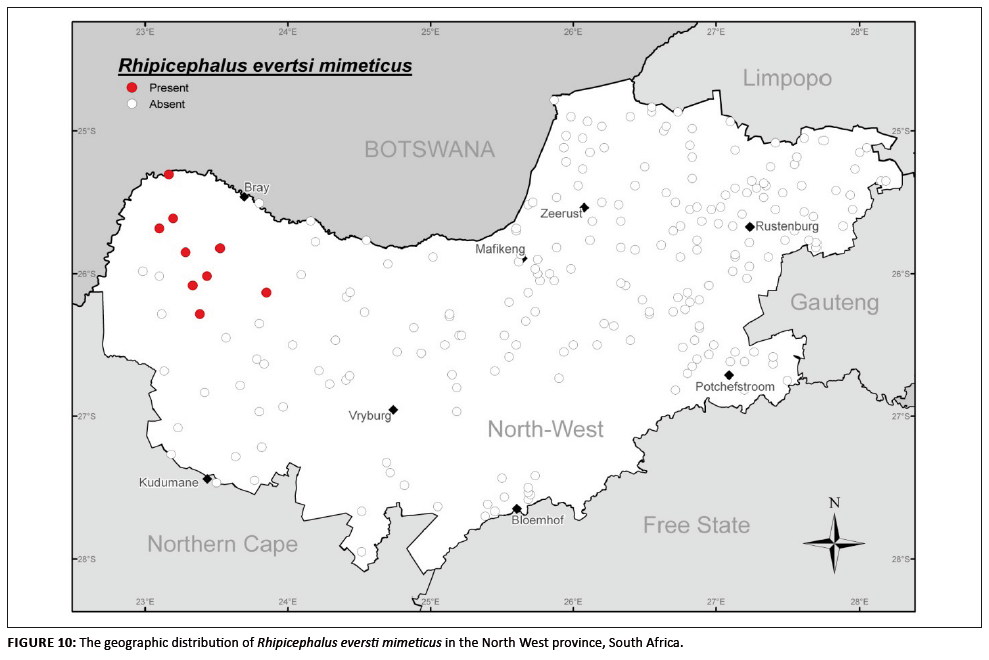 |
FIGURE 10: The geographic distribution of Rhipicephalus eversti mimeticus in the North West province, South Africa.
|
|
Rhipicephalus simus
This species establishes in regions with a savanna climate and is never encountered in high numbers (Norval & Mason 1981; Walker et al. 2000). It is
well established in the bushveld areas of the north-eastern region of the province to which its distribution is confined (Figure 11). Of the R. simus
collected, 71.9% were from cattle, 4.0% from sheep and 18.5% from goats. This species was also recovered from dogs (4.0%), whilst 1.6% were not assigned a host by
collectors.
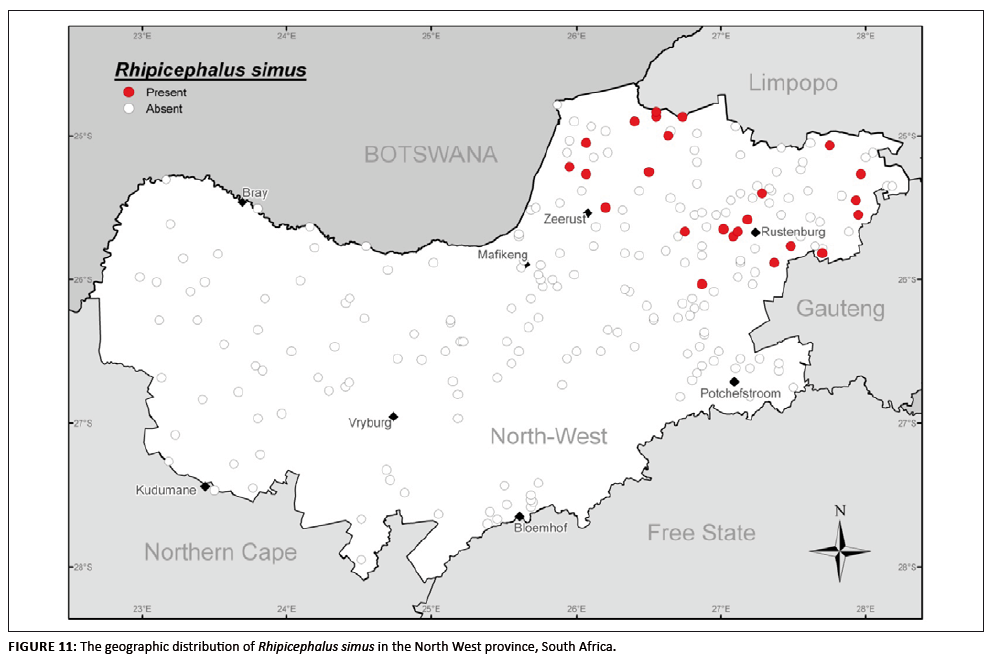 |
FIGURE 11: The geographic distribution of Rhipicephalus simus in the North West province, South Africa.
|
|
Tick-borne disease serology
As shown in Figures 7 and 8, R. (B.) decoloratus and R. (B.) microplus occurred sympatrically at some localities in the north-eastern region
of the province. Both species are associated with the transmission of the causative organism of anaplasmosis (Ana. marginale) as well as the organisms causing
bovine babesiosis (Babesia bigemina and Babesia bovis) (De Vos & Potgieter 1994). The antibody status of cattle herds to these tick-borne pathogens
in the areas of sympatric distribution of these two tick vector species, as well as the presence (or absence) of the two tick vector species is shown in Table 3.
|
TABLE 3: Serology results from livestock at various localities and associated presence or absence of vectors.
|
The results indicate that only one property (Commiesiedrift) is in a state of endemic stability (Norval et al. 1983) to B. bigemina (100%), whereby
sufficient infected vector ticks (R. [B.] decoloratus) are present to transmit the pathogen such that all the animals show an antibody response
indicative of disease immunity. All other properties appear to experience endemic instability to both B. bigemina and B. bovis, strongly suggesting that
intensive chemical control is practiced on these properties, thereby reducing vector challenge and, subsequently, pathogen transmission. On the endemically unstable
properties, more than 40% of the cattle show no antibodies to the causative organisms and are thus fully susceptible and at risk to babesiosis should they be
challenged by infected ticks.
None of the animals on six properties showed antibodies to B. bigemina despite the presence of the tick vector, possibly owing to uninfected ticks (unlikely)
or extremely stringent tick control practices. These animals could be completely susceptible to bovine babesiosis should they be challenged by infected ticks. R.
(B.) microplus was absent on five of these properties and, as expected, none of the animals tested positive for B. bovis antibodies. However, on one of
the properties (Leeukraal) on which no animals tested positive for B. bigemina in the presence of the tick vector, 50% of the animals tested positive for B.
bovis in the presence of the vector tick. The latter case is difficult to explain: it is possibile that R. (B.) microplus, as a vector, presented a
much higher challenge than did R. (B.) decoloratus and that antibody manifestation to B. bigemina was subsequently lost. On two properties,
respectively 28% and 40% of the animals showed antibody titres to B. bovis, indicative of R. (B.) microplus infestation, although this vector
tick was not collected at the time of the survey.
This study was conducted to survey the occurrence and distribution of ticks infesting livestock in the North West province of South Africa. The survey entailed
the monthly collection of tick specimens from livestock hosts at specified sites in the north-eastern, central and western regions of the province. Tick specimens
were subsequently identified and the distributions of the major species plotted.
According to this survey, livestock in this province harbour 22 tick species (18 ixodids; 4 argasids). The major tick-borne diseases were prevalent mainly in the
north-eastern region, which also displayed the highest tick species diversity.
The vectors of Corridor disease (buffalo-associated Theileria parva), namely R. appendiculatus and Rhipicephalus zambeziensis were present in
the north-eastern region of the province, which indicates that care should be exercised in the introduction of Corridor-infected buffalo in these regions. The central
region appears transitory to the major vectors A. hebraeum and R. (B.) decoloratus, whilst the two Hyalomma vectors of Crimean–Congo
haemorrhagic fever virus are widespread over the whole province. The north-western area (Bray) of the western region has been infiltrated with R. evertsi
mimeticus, a species considered to be non-endemic to South Africa. Most herds sampled for serology in areas endemic for babesiosis and anaplasmosis in the
north-eastern region are endemically unstable and at risk to these tick-borne diseases should vector control measures become ineffective.
This study was funded by the Directorate of Veterinary Services of the North West province (Department of Agriculture). We wish to thank the Directorate for
their wholehearted and unstinting support for this survey, at managerial and operational level. Special thanks go to the many officers directly involved in the
collections.
Acocks, J.P.H., 1988, Veld types of South Africa with accompanying veld type map, 3rd edn., Department of Agriculture and Water Supply, Pretoria.
Apanaskevich, D.A. & Horak, I.G., 2008, ‘The genus Hyalomma Koch, 1844. V. Re-evaluation of the taxonomic rank of taxa comprising the
H. (Euhyalomma) marginatum group of species (Acari: Ixododidae) with redescription of all parasitic stages and notes on biology’,
International Journal of Acarology 34, 13−42. doi:10.1080/01647950808683704
Beati, L. & Keirans, J.E., 2001, ‘Analysis of the systematic relationships among ticks of the genera Rhipicephalus and Boophilus
(Acari: Ixodidae) based on mitochondrial 12S ribosomal DNA gene sequences and morphological characters’, Journal of Parasitology 87, 32−48.
doi:10.2307/3285173,
PMid:11227901
Coetzer, J.A.W. & Tustin, R.C., 2004, Infectious diseases of livestock, Oxford University Press, Cape Town.
De Castro, J.J., 1997, ‘Sustainable tick and tickborne disease control in livestock improvement in developing countries’, Veterinary Parasitology
71, 77−97. doi:10.1016/S0304-4017(97)00033-2
De Vos, A.J. & Potgieter, F.T., 1994, ‘Bovine babesiosis’, in J.A.W. Coetzer, G.R. Thomson & R.C. Tustin (eds.), Infectious diseases of
livestock with special reference to southern Africa, pp. 278–294, Oxford University Press, Cape Town.
Department of Agriculture, Forestry and Fisheries, 2004, Livestock Statistics 2004, Department of Agriculture, Forestry and Fisheries, Pretoria.
Gothe, R., 1999, Zeckentoxikosen [Tick toxicoses], Hieronymus Buchenproduktion, Munchen.
Guglielmone, A.A., Robbins, R.G., Apanaskevich, D.A., Petney, T.N., Estrada-Peña, A., Horak, I.G. et al., 2010, ‘The Argasidae, Ixodidae
and Nuttalliellidae (Acari: Ixodida) of the world: a list of valid species names’, Zootaxa 2528, 1−28.
Horak, I.G., Sheppey, K., Knight, M.M. & Beuthin, C.L., 1986, ‘Parasites of domestic and wild animals in South Africa. XXI. Arthropod parasites of vaal
ribbok, bontebok and scrub hares in the western Cape Province’, Onderstepoort Journal of Veterinary Research 53, 187−197.
Horak, I.G., MacIvor, K.M., Petney, T.N. & De Vos, V., 1987, ‘Some avian and mammalian hosts of Amblyomma hebraeum and Amblyomma marmoreum
(Acari, Ixodidae)’, Onderstepoort Journal of Veterinary Research 54, 397−403.
Horak, I.G., Camicas, J.-L. & Keirans, J.E., 2002, ‘The Argasidae, Ixodidae and Nuttalliellidae (Acari: Ixodida): a world list of valid tick names’,
Experimental and Applied Acarology 28, 27−54. doi:10.1023/A:1025381712339
Jongejan, F. & Uilenberg, G., 1994, ‘Ticks and control methods’, Revue Scientifique et Technique de Office International des Epizooties
13, 1201−1226.
Joyner, L.P., Donnely, J., Payner, R. & Brocklesby, D.W., 1972, ‘The indirect fluorescent antibody test for the differentiation of infections with
Babesia divergens or Babesia major’, Research in Veterinary Science 13, 515–518.
PMid:4567732
Mason, C.A. & Norval, R.A.I., 1980, ‘The ticks of Zimbabwe. I. The genus Boophilus’, Zimbabwe Veterinary Journal 11, 36−43.
Minjauw, B., 2005, ‘The economic impact of heartwater (Cowdria ruminantium infection) in the Republic of South Africa, and of its control through
the use of new inactivated vaccines’, Draft Country report, UF/USAID/SADC Heartwater Research Project, Washington, DC.
Murrell, A.N., Campbell, J.H. & Barker, S.C., 2000, ‘Phylogenetic analysis of the rhipicephaline ticks indicates that the genus Rhipicephalus
is paraphyletic’, Molecular Phylogenetics and Evolution 16, 1−7.
doi:10.1006/mpev.2000.0762,
PMid:10877935
Ndung’u, L.W., Aguirre, C., Rurangirwa, F.R., McElwain, T.F., McGuire, T.C., Knowles, D.P. et al., 1995, ‘Detection of Anaplasma ovis
infection in goats by major surface protein 5 competitive inhibition enzyme-linked immunosorbent assay’, Journal of Clinical Microbiology 33,
675–679.
Neitz, W.O., 1959, ‘Sweating sickness: the present state of our knowledge’, Onderstepoort Journal of Veterinary Research 23, 3−38.
Norval, R.A.I. & Mason, C.A., 1981, ‘The ticks of Zimbabwe. II. The life cycle, distribution and hosts of Rhipicephalus simus Koch, 1844’,
Zimbabwe Veterinary Journal 12, 2−9.
Norval, R.A.I. & Horak, I.G., 2004, ‘Vectors: Ticks’, in J.A.W. Coetzer & R.C. Tustin (eds.), Infectious diseases of livestock, 2nd edn.,
pp. 3−42, Oxford University Press, Cape Town.
Norval, R.A.I., Fivaz, B.H., Lawrence, J.A. & Daillecourt, T., 1983, ‘Epidemiology of tick-borne diseases of cattle in Zimbabwe. I. Babesiosis’,
Tropical Animal Health and Production 15, 87–94.
doi:10.1007/BF02239802,
PMid:6868134
Nyangiwe, N. & Horak, I.G., 2007, ‘Goats as alternative hosts of cattle ticks’, Onderstepoort Journal of Veterinary Research 74, 1−7.
PMid:17708147
Rechav, Y., 1982, ‘Dynamics of tick populations (Acari: Ixodidae) in the eastern Cape Province of South Africa’, Journal of Medical Entomology
19, 679−700. PMid:7154025
Spickett, A.M. & Heyne, H., 1988, ‘A survey of Karoo tick paralysis in South Africa’, Onderstepoort Journal of Veterinary Research 55,
89−92. PMid:3393373
Spickett, A.M. & Heyne, H., 1990, ‘The prehatch period and larval survival of Boophilus decoloratus (Koch, 1844) (Acarina, Ixodidae) under
natural conditions in the Transvaal, South Africa’, Onderstepoort Journal of Veterinary Research 57, 95−98.
PMid:2339003
Theiler, G., 1962, ‘The ixodoidea parasites of vertebrates in Africa south of the Sahara (Ethiopian Region), Project S 9958’, Report to
the Director of Veterinary Services, Onderstepoort.
Van Rensburg, S.J.J., 1981, ‘The importance and need for tick control – what it means to the industry’, in G.B. Whitehead & J.D. Gibson
(eds.), Tick biology and control: International conference proceedings, Grahamstown, South Africa, January 27–29, 1981, pp. 119–122.
Visser, E.S., McGuire, T.C., Palmer, G.H., Davis, W.C., Shkap, V., Pipano, E. et al., 1992, ‘The Anaplasma marginale msp5 gene encodes a
19-kilodalton protein conserved in all recognized Anaplasma species’, Infection and Immunity 60, 5139–5144.
PMid:1280624,
PMid:258289
Walker, A.R., Bouattour, J.-L., Camicas, A., Estrada-Pena, I.G., Horak, I.G., Latif, A.A. et al., 2003, Ticks of domestic animals in Africa: a guide
to identification of species, Bioscience reports, Edinburgh.
Walker, J.B., Keirans, J.E. & Horak, I.G., 2000, The genus Rhipicephalus (Acari, Ixodidae): a guide to the brown ticks of the world, Cambridge
University Press, Cambridge.
|
|
