|
Article Information
|
Authors:
Mmeta G. Yongolo1
Henrik Christensen2
Kurt Handberg3
Uswege Minga4
John E. Olsen2
Affiliations:
1Department of Virology, Ministry of Livestock Development, Tanzania
2Department of Veterinary Disease Biology, University of Copenhagen, Denmark
3Department of Clinical Microbiology, University Hospital of Aarhus, Denmark
4Faculty of Science, Technology and Environmental Studies, Open University of Tanzania, Tanzania
Correspondence to:
Henrik Christensen
Email:
hech@life.ku.dk
Postal address:
Department of Veterinary Disease Biology, University of Copenhagen, Stigbøjlen 4, 1870 Frederksberg C, Denmark
Dates:
Received: 20 Dec. 2010
Accepted: 08 Apr. 2011
Published: 30 Sept. 2011
How to cite this article:
Yongolo, M.G., Christensen, H., Handberg, K., Minga, U. & Olsen, J.E., 2011, ‘On the origin and diversity of Newcastle disease virus in Tanzania’,
Onderstepoort Journal of Veterinary Research 78(1), Art. #312, 8 pages.
doi:10.4102/ojvr.v78i1.312
Copyright Notice:
© 2011. The Authors. Licensee: AOSIS OpenJournals. This work
is licensed under the Creative Commons Attribution License.
ISSN:
0030-2495 (print)
ISSN:
2219-0635 (online)
|
|
|
|
On the origin and diversity of Newcastle disease virus in Tanzania
|
|
In This Original Research...
|
Open Access
|
• Abstract
• Introduction
• Materials and methods
• Materials
• Viral RNA extraction
• Primers and reverse transcriptase polymerase chain reaction
• Sequence analysis, alignment and phylogenetic analysis
• Results
• Discussion
• Conclusion
• References
|
|
Free-range rural chickens (FRCs) dominate the poultry industry in developing countries and chickens are exposed to multi-host infections, including Newcastle
disease virus (NDV). The knowledge about the characteristics of NDV from FRCs is limited. This study investigated the persistence, spread and risks of
NDV from FRCs. NDV isolates (n = 21) from unvaccinated FRCs in Tanzania were characterised by conventional intracerebral pathogenicity index (ICPI) and
sequence analysis of a partial region of the deduced fusion protein encompassing the cleavage site. Results showed that five isolates were screened as lentogenic,
nine as mesogenic and six as velogenic. Phylogenetic analysis of the 21 isolates compared to reference sequences revealed three, four, nine and five isolates in g
enotypes 1, 2, 3c and 4a, respectively. Genotype 3c also included published sequences of Tanzanian isolates obtained from exotic birds and chicken isolates from
Uganda. The analysis showed that NDV were persistently present among chicken populations and possibly spread through live chicken markets or migration of wild
birds. Differences in amino acid sequences detected around the cleavage site separated the isolates in six types. However, cleavage site pattern could not fully
differentiate mesogenic isolates from velogenic isolates.
Free-range rural chickens (FRCs) populations dominate the poultry industry in developing countries (Permin & Hansen 1998; Regmi 2001). The management of FRC
allows interactions between chickens and ducks, pigeons, wild birds and other animals. This predisposes chickens to multi-host infections, including Newcastle
disease virus (NDV). Moreover, FRC flocks and populations are composed of multi-age chickens, which are usually not vaccinated. Over 250 species of birds are
reported to be susceptible to NDV infection (Alexander 2001). Chickens are the most susceptible species and infections of susceptible birds with virulent NDV
isolates lead to severe disease and heavy stock losses (Alexander 2001). Thus, Newcastle disease (ND) is apparently responsible for major losses in FRC populations
(Spradbrow 1993). It is also of economical importance in commercial poultry production and trade worldwide (Alexander 2001). Major outbreaks have been reported since
ND was reported for the first time (Lomniczi et al. 1998). Newcastle disease occurs sporadically among FRCs, with seasonal epidemics and up to 100% mortality
in unvaccinated flocks (Spradbrow 2000; Yongolo, Machangu & Minga 2002). Whereas much has been reported on NDV contributing to outbreaks in commercial production,
knowledge is limited about the NDV circulating and causing ND oubreaks in FRC populations.
NDV are categorised as avian paramyxovirus serotype 1 (APMV-1) and belongs to the genus Avulavirus, subfamily Paramyxovirinae in the family Paramyxoviridae
(eds. Van Regenmortel et al. 2000). The APMV-1 serotype has a negative-sense, single-stranded RNA genome composed of six genes coding for RNA-directed RNA
polymerase, haemagglutinin-neuraminidase protein, fusion protein (F), matrix protein, phosphoprotein and nucleoprotein (Philips, Samson & Emmerson 1998).
Fusion protein synthesised as a precursor F0 must be proteolytically cleaved to the peptides F1 and F2 for fusion activity to occur
between virus and host. This separation occurs between amino acid residues 116 and 117, with postion 117 being the first amino acid for the F1 fusion
polypeptide. The sequence 111GGRQGR|L117 of an avirulent isolate turned virulent when converted to 111GRRQRR|F117 (Peeters
et al. 1999) and the peptide sequence at the cleavage site can be used to predict virulence.
Virulence of NDV is conventionally estimated by determining the intracerebral pathogenicity index (ICPI), intravenous pathogenicity index and the mean death time in
day-old chicks, 6-week-old chicks and embryonated chicken eggs (ECEs) of between 9 and 11 days old, respectively (Office International des Epizooties 2009). Results
from these three methods cannot differentiate between strains and used alone they have very little epidemiological significance. Pathotyping by such methods can only
classify NDV a highly (velogenic), moderately (mesogenic) and slightly (lentogenic) pathogenic. However, strain differentiation and epidemiological data can be
achieved through restriction enzyme analysis, cleavage site mapping and sequence analysis of the fusion protein gene (Ballagi-Pordány et al. 1996).
Based on these methods, eight genotypes have been revealed to have been involved in major epidemics (Yu et al. 2001). The genotypes have recently been
reclassified and 16 new genotypes are now recognised (Aldous et al. 2003). Of the recent genotypes, novel genotypes VIIb, VIId and VIII (5b, 5d and 3d according
to Aldous et al. 2003) were identified from outbreaks in southern Africa, Asia and Europe (Abolnik et al. 2004; Herczeg et al. 1999; Li
et al. 2001; Lomniczi et al. 1998; Yang et al. 1999).
In this study, 21 NDV isolates from Tanzanian FRCs and ducks, with no history of vaccination against ND (Yongolo 1996), were characterised to gain molecular
epidemiological information. However, the characteristics of NDV affecting FRCs that could influence endemicity were not addressed or documented (Yongolo 1996).
The aim of this study was to demonstrate NDV persistence, spread and infection FRC to further understanding of the epidemiology of ND in FRCs and possible risks to
the commercial poultry industry.
Materials
The sources of virus are shown in Table 1. These isolates were samples taken from Tanzanian FRCs between 1994 and 1995 at monthly intervals. They were confirmed
and deposited at the Central Veterinary Laboratories Agency, Weybridge, Surrey (UK) and deposited at the Sokoine University of Agriculture, Morogoro, Tanzania
(Yongolo 1996). All isolates were passaged three times in ECEs (between 9 and 11 days old) before use. Of the 21 isolates, one had been isolated from live chicken
markets in the Mbeya region of Tanzania and the rest from village chicken in the regions Morogoro and Tabora. One isolate was obtained from a duck kept with chickens
in the same flock (Figure 1).
|
TABLE 1: List of Newcastle disease virus isolates investigated.
|
 |
FIGURE 1: Map of Tanzania with indication of the three regions from where
isolates were received.
|
|
Before RNA extraction, all isolates were passaged in 10-day-old ECEs and the presence of NDV in the allantoic harvests was detected following standard procedures
of haemagglutination (HA) and HA inhibition test according to the Commission of the European Communities (1992). The virulence of each isolate
after the third passage was estimated by determining the ICPI in day-old chicks according to standards of the Office International des Epizooties (OIE 2009).
Viral RNA extraction
Viral RNA was extracted directly from the allantoic fluid harvested from NDV-infected ECEs. Virus extraction was performed at the Danish Veterinary Laboratory
in Aarhus, Denmark. RNA was isolated using the QIAgen RNeasy Mini Kit RT-PCR system (QIAgen, Hilden, Germany) and Expand (Roche Diagnostics, Hvidovre, Denmark)
according to the protocol provided with the QIAgen RNeasy® kit. Cells in 400 µL of the allantoic fluid were lysed and DNase inactivated using 284 µL
RTL buffer that contained guanidine isothiocyanate (GITC), with subsequent addition of 6 µL of β-mercaptoethanol. The spin column was washed using the two
washing buffers, RW1 and RPE, provided in the kit and followed by centrifugation. Elution was performed with 50 µL RNase-free water and the elute was preserved
at –20 °C until use.
Primers and reverse transcriptase polymerase chain reaction
Primer sequences, originally designed to amplify the F protein gene between position 148 and 535 to yield an expected product 388 nucleotides long, were obtained
from Jørgensen et al. (1999). The procedure for single-tube reverse transcriptase polymerase chain reaction (RT-PCR) was carried out according to
Jørgensen et al. (1999) using MgCl2at a concentration of 1.5 mM. All reagents were handled on ice and the enzyme was added just before
transferring the reagents to the PCR machine (PTC-200, MJ Research, Watertown, Massachusetts). Initial reverse transcription was performed for 30 min at 50 °C
and denaturation was then carried out for 2 min at 94 °C. Ten cycles, entailing denaturation for 30 s at 94 °C, annealing for 30 s at 58 °C and elongation
for 1 min at 68 °C, were performed. After 10 cycles, an additional 25 were performed with the elongation time extended by 5 s in each cycle. Finally, elongation
was performed for 7 min at 68 °C.
After RT-PCR, DNA was extracted from a sliced agarose gel (stained with ethidium bromide) using the QIAgen II Gel Extraction kit as described by the QIAEX II Handbook
(QIAgen, Hilden, Germany). The elute was centrifuged and the DNA-containing supernatant was stored at –20 °C until use. Sequencing of the RT-PCR-derived
nucleotide sequences was determined by cycle sequencing (Sears et al. 1992), using the Amplitaq FS dye terminator kit and the 373A automatic sequencer
(Applied Biosystems, Foster City, California). The primers NDV-2 and NDV-3 (Jørgensen et al. 1999) used during PCR amplification were also used in
sequencing. The conditions for performing DNA sequencing and data evaluation were as recommended by Applied Biosystems.
Sequence analysis, alignment and phylogenetic analysis
Sequence analysis was performed on 17 isolates from FRCs and a duck. Sequences were compared to reference sequences from other parts of the world, selected to
represent the 16 genotypes reported to date (Aldous et al. 2003). Further selection of sequences was based on the BLAST search programme (Altschul et al.
1997) in the GenBank database (Benson et al. 2004) in order to identify closely related sequences. Only sequences with clear indication of isolate identity, host
and country of origin were included. Six sequences previously determined from Tanzanian isolates (Aldous et al. 2003; Seal et al. 1998) and all sequences
described for the African continent were also included. The region of the F protein gene between position 231 and 374 (B1 isolate, accession number NC_002617) was
aligned with representatives of published genotypes resulting in 180 positions for comparison. Pair-wise sequence comparisons were performed using EMBOSS (WATER)
(http://www.ebi.ac.uk). The phylogenetic analysis was perfomed by neighbour joining analysis by ClustaxX
(Thompson et al. 1997) and the tree visualised by MEGA4 (Tamura et al. 2007).
Results from the virulence assessment in day-old chicks showed the ICPI to be between 0.0 and 2.0 (Table 1). ICPI intervals of 0.00–0.70, 0.71–1.20 and
1.21–2.00 were used to classify the virus as lentogenic, mesogenic or velogenic, respectively.
The phylogenetic tree is shown in Figure 2. NDV isolates from Tanzania were asigned to four of the 16 genotypes defined earlier (Aldous et al. 2003). The
three isolates classified as genotype 1 (Table 2 and Figure 2) showed 98.4–99.2% nucleotide similarity. Four isolates, including the only one from the Mbeya
region and the reference strain B1, were described as belonging to genotype 2 (Table 2 and Figure 2). The isolates of this group investigated in the current study
showed 97.4–99.6% DNA sequence similarity. BLAST search showed nearly 100 sequences with high similarity to this genotype 2, including more versions of the
LaSota strain (data not shown).
|
TABLE 2: Protein signatures of the F gene cleavage site of Newcastle disease virus compared to published cleavage sites of slightly virulent and virulent Newcastle disease virus.
|
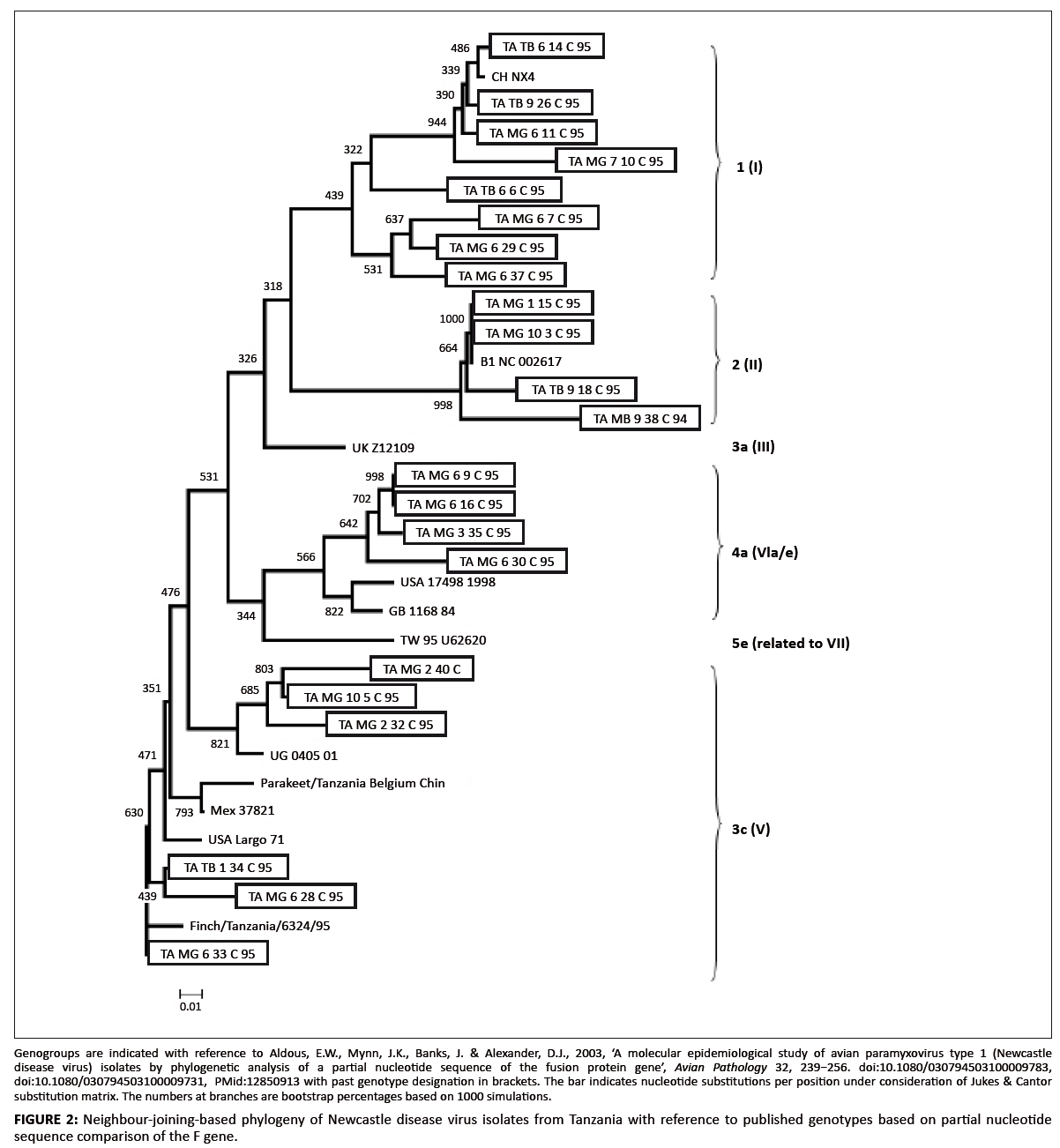 |
FIGURE 2: Neighbour-joining-based phylogeny of Newcastle disease virus isolates from Tanzania with reference to published genotypes based on partial nucleotide
sequence comparison of the F gene.
|
|
Nine isolates belonged to genotype 3c (Table 1 and Figure 2). The isolates showed 90.6–100% DNA sequence similarity. Isolates MG6.33C and TB1.34 even shared
sequences, as did MG10.4C and MG10.5C (Table 2). The only duck isolate was included in this genotype. The Ugandan outbreak strain also belonged to this group and
showed the highest similarity to isolate MG10.5C (97.9%). The presence of identical sequences in different regions of Tanzania, as well as the high similarity to
the strain from Uganda, indicates rapid spread of NDV. Isolate 6324/95 sampled in 1995 from wild birds represented in the investigation of Seal et al. (1998)
also belonged to this group.
Five Tanzanian isolates were grouped as belonging to genotype 4a (Table 2 and Figure 2). The isolates showed 96.6–99.0% DNA sequence similarity. Isolate MG3.35C
was isolated in March 1995, while MG6.9C, MG6.16C and MG6.30C were isolated in June 1995. MG3.35C may have been the progenitor NDV for the June isolates. This group
showed only little similarity to other sequences deposited in the nucleotide databases (< 92%), as determined by the BLAST search.
The predicted amino acid sequences of the fusion protein gene region (positions 231–420) were aligned. The variations observed for the Tanzanian isolates are
shown in Table 2. All isolates for genotypes 1 and 2 showed leucine at position 117 of the cleavage site, whereas genotypes 3c and 4a sequences showed phenylalanine
at that position.
According to Aldous and Alexander (2001) NDV is an APMV-1 with an ICPI of 0.7 or higher or with at least three basic amino acids between positions 113 and 116 and
phenylalanine at position 117. Most of the APMV-1 virus isolated in the present study fit this definition, except for three isolates (MG10.3C, MG7.10C, TB9.26C)
with an ICPI of 0.5 or lower and leucine at position 117 (see Tables 1 and 2).
The NDV isolates from Tanzania could be classified as genotypes 1, 2, 3c and 4a according to the Aldous et al. (2003) designations. Genotype 2 viruses are
known to be vaccine strains mainly isolated from the USA (Herczeg et al. 1999; Liang et al. 2002). The LaSota strain, which is the commonly used type
of vaccine in Tanzania, could have been the progenitor virus for some of the isolates involved in disease.
The study showed that isolates belonging to genotype 3c were dominant and involved in ND outbreaks amongst FRCs in Tanzania and probably also other east African
countries. The nine isolates were velogenic or mesogenic and isolated from apparently healthy or sick birds (Table 1). Otim et al. (2004) identified a member
of genotype 3c from ND outbreaks in Uganda, which may be related to those from Tanzania in the current investigation. This would confirm that poultry move between
Uganda and Tanzania. However, other sources for these isolates may also exist, because Lomniczi et al. (1998) reported that old Hungarian isolates
belong to this group. Members of this group have also been reported in the USA, Mexico (see Figure 2) and Germany, dating back to the 1970s (Wehmann et al.
2003). The isolates reported by Seal et al. (1998) from finch and parakeet also belonged to this group.
Unfortunately, Abolnik et al. (2004) sequenced another partial region than we did and sequences could therefore not be compared phylogenetically. Based on the
comparison of labelled groups, group 3c (old V) was found in Tanzania but does seem to have been present in South Africa. Groups IV, VIIb, VIIc, VIId and VIII, in turn,
were found in South Africa but not in Tanzania.
The presence of genotype 1 NDV was reported in Tanzania for the first time. It is apparent from this study that genotype 1 NDV are still prevalent. Members of genotype
1, together with those of genotypes 2, 3a and 3b (Aldous et al. 2003), have been reported to be involved in the first panzootic outbreaks (Herczeg et al.
1999; Yu et al. 2001). Because genotype 1 NDV were involved in the first panzootic outbreak, which started in the mid-1920s in other parts of the world, and the
recovery of related NDV in Tanzania in 1995, old genotypes are possibly still circulating worldwide, together with new genotypes. It is well known from previous
findings that genotype 1 viruses are mainly lentogenic (Collins, Bashiruddin & Alexander 1993; Peeters et al. 1999; King & Seal 1998). All three genotype
1 isolates had leucine at position 117, which is found only in low-virulence NDV (Collins, Govey & Alexander 2003; Kant et al. 1997; Oberdörfer &
Werner 1998; Seal, King & Bennett 1995), whereas all virulent NDV have phenylalanine at position 117 (Collins et al. 1993). Genotype 1 comprised
representative isolates from the two sampled regions of Tanzania. It is not known when genotype 1 viruses could have been introduced into Tanzania. Isolate V4 could
possibly have been introduced into Tanzania as a thermostable vaccine for research purposes in 1992, 1995 and 1996 (Foster et al. 1999). Given the geographical
location and the short time since V4 was introduced in Tanzania it is most unlikely that it is the progenitor virus for this group. On the other hand, V4 may have
spread from neighbouring Malawi, where extensive field trials using the thermostable V4 had been carried out to control ND in intensive and smallholder village chicken
flocks before 1987 (Sagild & Haresnape 1987).
The absence of genotypes 5a, 5b, 5c, 5d, 3d (Aldous et al. 2003) viruses in Tanzania indicates that NDV, which caused devastating outbreaks of velogenic ND from
1993 to 1995 in southern African countries, did not reach Tanzania or were not isolated (Herczeg et al. 1999). Recent outbreaks from Nigeria were found to belong
to genotypes 5f/g/h and 3a in addition to 1 and 2 (Snoeck et al. 2009); however, members of these groups were not identified from Tanzania. We can conclude that
no particular unique types of NDV exist in Tanzania, but that the current types observed probably have descended from past outbreak strains in other parts of the world
as well as from vaccine strains.
No temporal correlation was found between isolates sampled in Morogoro, Tabora or Mbeya. At Morogoro and Tabora all four genotypes were sampled; however, most isolates
were obtained from Morogoro in June 1995 and they belonged to three genotypes (1, 3c and 4a). The study has shown that NDV is endemic to Tanzania, because NDV was
isolated from village and markets in different geographical locations at different times of the year. Our study also showed that the different isolates from the chickens
and ducks showed different levels of virulence and that multiple lineages were circulating amongst domestic birds. This is important because such birds could be the
source of potentially virulent NDV to FRCs and commercial poultry worldwide. The presence of different pathotypes was confirmed by ICPI and the presence of variable
amino acid sequences at the cleavage site. The presence of variable pathotypes in the field may complicate disease outcome and diagnosis. Therefore, mere isolation of
NDV cannot be used for ND diagnosis or for determining the prevalence of ND unless characterisation and clinical disease are correlated. The persistent presense of NDV
in FRCs is important because they predispose chickens to secondary and opportunistic upper respiratory bacterial infections (such as from Escherichia coli).
Therefore, frequent programmed vaccination is recommended. This will guarantee protection of most chickens in an endemic situation. Enforcement of biosecurity may be
the best alternative, but this is not feasible in the free-range system with its low input and low output management system, as is commonly practiced in Tanzania and
other developing countries.
Genetic variations amongst NDV in FRCs were established in this study. These results showed an epidemiological relationship between NDV from FRCs of Tanzania to NDV
reported in other countries. Accorrding to Tsai et al. (2000), viral divergence of 0.2–0.9% indicates that the viruses are genetically highly homogenous
and similar. In the present study, more than 99% similarity was found amongst some members of all genotypes and even between isolates from different regions (Morogoro
and Tabora). DNA similarity of up to 100% amongst some isolates found in the two regions studied and the phylogenetic tree analysis provided evidence that the isolates
had a common origin. However, results in this study showed that no group of viruses was found exclusively in Tabora, although a specific group of viruses was found
only in Morogoro. Isolates from Tabora may therefore have had the same ancestor as some of the Morogoro isolates. The one-way type of relationship is attributed to the
one-way marketing of live chickens from western regions to coastal regions of Tanzania. NDV could have spread from Tabora to Morogoro through live bird marketing,
which is commonly practised (Mlozi et al. 2003). Therefore, live bird marketing may be the main vehicle for spreading ND in Tanzania.
Several amino acid sequence differences were detected in the studied isolates, specifically differences with regard to the fusion protein cleavage site. The differences
correlated with virulence. Most results showed correlation between genotype F cleavage sequence and virulence for both slightly virulent and virulent NDV isolates.
These results are in agreement with what was reported by Yu et al. (2001). Lack of correlation between ICPI values and the cleavage site motif, together with the
findings on amino acid substitutions around the cleavage site, which leads to NDV with changed virulence, means that the actual virulence characteristics of an isolate
cannot be determined by the cleavage site motif alone. Consequently, analysis of cleavage site motifs can be used only qualitatively to determine the virulence of NDV
isolates. Therefore, the ICPI cannot be replaced in totality by analysis of the cleavage site. Furthermore, these results show that fusion protein gene cleavage site
is not the only factor that determines virulence. Just as for the Avian influenza virus (Alexander 2000; Tollis & Di Trani 2002) HN protein precursor also plays a
role in the virulence of NDV (Cobaleda et al. 2002) and findings by Huang et al. (2003) show V protein of NDV significantly contribute to the virus
virulence as an alpha/beta interferon antagonist. Thus, although F cleavage site is the major determinant of virulence, other factors also contribute to actual virus
virulence.
Changes of NDV from avirulent to virulent was previously suspected from two outbreaks of ND amongst poultry in Ireland (Alexander et al. 1992). The causative NDV
were velogenic and very similar, antigenically and genetically, to avirulent viruses isolated from feral waterfowls, suggesting that velogenic viruses arose from
avirulent NDV from wild birds (Collins et al. 1993; Collins et al. 1998). Furthermore, recent outbreaks in Australia (1998–2001) revealed genetic
resemblance of virulent NDV to field lentogenic isolates (Gould et al. 2001). In both field observations and the recent experimental study by Shengquing et
al. (2002), step-by-step amino acid substitution was demonstrated. The management of FRCs allows for unlimited contact with other hosts including wild birds found
to be infected mainly with lentogenic NDV, with the exception of few species (Takakuwa et al. 1998). This means that avirulent and lentogenic NDV isolates with
avirulent genes have the potential to become velogenic after transmission to and repeated passage in chickens. Exposure to NDV and repeated passage are highest in FRCs
compared to commercial chickens. Therefore, control of ND in FRC populations is important to reduce the chances of generating unselected populations of different NDV.
The presence of isolates in FRCs with drifting virulence suggests that virulent isolates could emerge from lentogenic vaccine viruses. These results have important
implications for the use of live virus vaccine in the control of ND amongst rural FRCs as well as commercial chickens. Although there are no reports of cases of
virulent NDV arising from the current live vaccines, future plans about ND control should take the possibility into consideration, especially with regard to ND control
in rural FRC populations. It is recommended that old and new vaccines should be carefully selected and, when introduced, monitored to detect their conversion to
virulence.
In conclusion, investigation of NDV isolated from FRCs in Tanzania showed presence of lentogenic, mesogenic and velogenic types, which
phylogenetic analysis of a partial region of the fusion protein gene allocated to genotypes 1, 2, 3c and 4a. Genotype 3c also included published sequences of
Tanzanian isolates obtained from exotic birds and chicken isolates from Uganda. The analysis showed that NDV were persistently present among chicken populations
and possibly spread through live chicken markets or migration of wild birds.
Abolnik, C., Horner, R.F., Bisschop, S.P., Parker, M.E., Romito, M. & Viljoen, G.J.A., 2004, ‘A phylogenetic study of South African Newcastle disease
virus strains isolated between 1990 and 2002 suggests epidemiological origins in the Far East’, Archives of Virology 49, 603−619.
doi:10.1007/s00705-003-0218-2,
PMid:14991446
Aldous, E.W. & Alexander, D.J., 2001, ‘Detection and differentiation of Newcastle disease virus (avian paramyxovirus type 1)’, Avian
Pathology 30, 117–128. doi:10.1080/03079450120044515,
PMid:19184885
Aldous, E.W., Mynn, J.K., Banks, J. & Alexander, D.J., 2003, ‘A molecular epidemiological study of avian paramyxovirus type 1 (Newcastle disease virus)
isolates by phylogenetic analysis of a partial nucleotide sequence of the fusion protein gene’, Avian Pathology 32, 239−256.
doi:10.1080/030794503100009783,
doi:10.1080/0307945031000097831,
PMid:12850913
Alexander, D.J., 2000, ‘A review of avian influenza in different bird species’, Veterinary Microbiology 74, 3−13.
doi:10.1016/S0378-1135(00)00160-7
Alexander, D.J., 2001, ‘Gordon Memorial Lecture: Newcastle Disease’, British Poultry Science 42, 5−22.
doi:10.1080/713655022,
PMid:11337967
Alexander, D.J., Campbell, G., Manvell, R.J., Collins, M.S., Parsons, G. & McNulty, M.S., 1992, ‘Characterisation of an antigenically unusual virus
responsible for two outbreaks of Newcastle disease in the Republic of Ireland in 1990’, Veterinary Records 130, 65−68.
doi:10.1136/vr.130.4.65,
PMid:1532467
Altschul, S.F., Madden, T.L., Schaffer, A.A., Zhang, J., Zhang, Z., Miller, W. et al., 1997, ‘Gapped BLAST and PSI-BLAST: a new generation of protein
database search programs’, Nucleic Acids Research 25, 3389−3402.
doi:10.1093/nar/25.17.3389,
PMid:9254694,
PMid:146917
Ballagi-Pordány, A., Wehman, E., Herczeg, J., Belák, S. & Lomniczi, B., 1996, ‘Identification and grouping of Newcastle disease virus
strains by restriction site analysis of a region from the F gene’, Archives of Virology 141, 243−261.
doi:10.1007/BF01718397
Benson, D.A., Karsch-Mizrachi, I., Lipman, D.J., Ostell, J. & Wheeler, D.L., 2004, ‘GenBank: update’, Nucleic Acids Research 32, D23−D26.
doi:10.1093/nar/gkh045,
PMid:14681350
PMid:308779
Commission of the European Communities (CEC). European Commission. 1992, ‘Legislation Governing: I The Control and Eradication of
Newcastle Disease and Avian Influenza. II Trade in Poultry, Poultry Meat and Hatching Eggs’. In: European Commission Directorate General VI. Agriculture.
Council Directive 92/66/EEC of 14 July 1992, Official Journal of the European Communities 5, L 260/1–L 260/20, viewed n.d., from
http://europa.eu/legislation_summaries/food_safety/animal_health/l12021_en.htm
Cobaleda, C., Munoz-Barroso, I., Sagrera, A. & Villar, E., 2002, ‘Fusogenic activity of reconstituted Newcastle disease virus envelope: a role for the
haemagglutinin-neuraminidase protein in the fusion process’, International Journal of Biochemistry and Cell Biology 34, 403−413.
doi:10.1016/S1357-2725(01)00127-3
Collins, M.S., Bashiruddin, J.B. & Alexander, D.J., 1993, ‘Deduced amino acid sequences at the fusion protein cleavage site of Newcastle disease viruses
showing variations in antigenicity and pathogenicity’, Archives of Virology 128, 363−370.
doi:10.1007/BF01309446,
PMid:8435046
Collins, M.S., Franklin, S., Strong, I., Meulemans, G. & Alexander, D.J., 1998, ‘Antigenic and phylogenetic studies on a variant Newcastle disease virus
using anti-fusion protein monoclonal antibodies and partial sequencing of the fusion protein gene’, Avian Pathology 27, 90−96.
doi:10.1080/03079459808419279,
PMid:18483969
Collins, M.S., Govey, S.J. & Alexander, D.J., 2003, ‘Rapid in vitro assessment of the virulence of Newcastle disease virus isolates using the ligase
chain reaction’, Archives of Virology 148, 1851−1862.
doi:10.1007/s00705-003-0131-8,
PMid:14505095
Foster, H.A., Chitukuro, H.R., Tuppa, E., Mwanjala, T. & Kusila, C., 1999, ‘Thermostable Newcastle disease vaccines in Tanzania’, Veterinary
Microbiology 68, 127−130. doi:10.1016/S0378-1135(99)00068-1
Gould, A.R., Kattenbelt, J.A., Selleck, P., Hansson, E., Della-Porta, A. & Westbury, H.A., 2001, ‘Virulent Newcastle disease in Australia: Molecular
epidemiology analysis of viruses isolated prior to and during the outbreaks of 1998–2000’, Virus Research 77, 51−60.
doi:10.1016/S0168-1702(01)00265-9
Gould, A.R., Hansson, E., Selleck, K., Kattenbelt, J.A., MacKenzie, M. & Della-Porta, A.J., 2003, ‘Newcastle disease virus fusion and
haemagglutinin-neuraminidase gene motifs as markers for viral lineage’, Avian Pathology 32, 361−373.
doi:10.1080/0307945031000121112,
PMid:17585460
Herczeg, J., Wehmann, E., Bragg, R.R., Travassos Dias, P.M., Hadjiev, G., Werner, O. et al., 1999, ‘Two novel genetic groups (VIIb and VIII)
responsible for recent Newcastle disease outbreaks in Southern Africa, one (VIIb) of which reached Southern Europe’, Archives of Virology 144,
2087−2099. doi:10.1007/s007050050624,
PMid:10603164
Huang, Z., Krishhamurthy, S., Panda, A. & Samal, S., 2003, ‘Newcastle disease virus V protein is associated with viral pathogenesis and functions as
an alpha interferon antagonist’, Journal of Virology 77, 8676–8685.
doi:10.1128/JVI.77.16.8676-8685.2003,
PMid:12885886,
PMid:167241
Jørgensen, P.H., Handberg, K.J., Ahrens, P., Hansen, H.C., Manwell, R.J. & Alexander, D.J., 1999, ‘An outbreak of Newcastle disease in free-living
pheasants (Phasianus colchicus)’, Journal of Veterinary Medicine B 46, 381–387.
doi:10.1046/j.1439-0450.1999.00243.x
Kant, A., Koch, G., Van Roozelaar, D.J., Balk, F. & Huurne, A.T., 1997, ‘Differentiation of virulent and non-virulent strains of Newcastle disease virus
within 24 hours by polymerase chain reaction’, Avian Pathology 26, 837−849.
doi:10.1080/03079459708419257,
PMid:18483949
King, D.J. & Seal, B.S., 1998, ‘Biological and molecular characterization of Newcastle disease virus (NDV) field isolates with comparisons to reference
NDV strains’, Avian Diseases 37, 961−969. doi:10.2307/1591900,
PMid:8141755
Li, Z., Nestor, K.E., Saif, Y.M., Anderson, J.W. & Patterson, R.A., 2001, ‘Effect of selection for increased body weight in turkeys on lymphoid organ
weights, phagocytosis, and antibody responses to fowl cholera and Newcastle disease-inactivated vaccines’, Poultry Science 80, 689−694.
Liang, R., Cao, D.J., Li, J.Q., Chen, J., Guo, X., Zhuang, F.F. & Duan, M.X., 2002, ‘Newcastle disease outbreaks in western China were caused by the
genotypes VIIa and VIII’, Veterinary Microbiology 87, 193−203.
doi:10.1016/S0378-1135(02)00050-0
Lomniczi, B., Wehmann, E., Herczeg, J., Ballagi-Pordány, A., Kaleta, E.F., Werner, O. et al., 1998, ‘Newcastle disease outbreaks in recent
years in Western Europe were caused by an old (VI) and a novel genotype (VII)’, Archives of Virology 143, 49−64.
doi:10.1007/s007050050267,
PMid:9505965
Mlozi, M.R.S., Kakengi, A.M.V., Minga, U.M., Mtambo, A.M. & Olsen, J.E., 2003, ‘Marketing of free-range local chicken in Morogoro and Kilosa urban
markets, Tanzania’, Livestock Research for Rural Development 15(2), viewed 28 May 2011, from
http://ftp.sunet.se/wmirror/www.cipav.org.co/lrrd/lrrd15/2/mloz152.htm
Oberdörfer, A. & Werner, O., 1998, ‘Newcastle disease virus: detection and characterisation by PCR of recent German isolates differing in
pathogenicity’, Avian Pathology 27, 237–243.
doi:10.1080/03079459808419330,
PMid:18483993
Office International des Epizooties (OIE), 2009, ‘Newcastle disease’, in Manual of Standards for Diagnostic Tests and Vaccines, 4th edn.,
viewed 28 May 2011, from
http://www.oie.int/fileadmin/Home/eng/Animal_Health_in_the_World/docs/pdf/NEWCASTLE_DISEASE__FINAL.pdf
OIE. See Office International de Epizooties.
Otim, M.O., Christensen, H., Jørgensen, P.H., Handberg, K.J. & Bisgaard, M., 2004, ‘Molecular characterization and phylogenetic study of
Newcastle disease virus isolates from recent outbreaks in Eastern Uganda’, Journal of Clinical Microbiology 42, 2802−2805.
doi:10.1128/JCM.42.6.2802-2805.2004,
PMid:15184476,
PMid:427882
Peeters, B.P.H., De Leeuw, O.S., Koch, G. & Gielkens, A.L., 1999, ‘Rescue of Newcastle disease virus from cloned cDNA: evidence that cleavability of the
fusion protein is a major determinant for virulence’, Journal of Virology 73, 5001–5009.
PMid:10233962,
PMid:112544
Permin, A. & Hansen, J.W., 1998, ‘Epidemiology, diagnosis and control of poultry parasites’, in FAO Animal Health Manuals 4. FAO, Rome.
Phillips, R.J., Samson, A.C.R. & Emmerson, P.T., 1998, ‘Nucleotide sequence of the 5 – terminus of Newcastle disease virus and assembly of the
complete genomic sequence: agreement with the “rule of six”’, Archives of Virology 143, 1993–2002.
doi:10.1007/s007050050435,
PMid:9856086
Regmi, A., 2001, ‘Changing structure of global food consumption and trade’, Economic Research Service, US Department of Agriculture and Trade,
Economic Research Service, Washington DC.
Sagild, I.K. & Haresnape, J.M., 1987, ‘The status of Newcastle disease and the use of V4 vaccine in Malawi’, Avian Pathology 16, 165−176.
doi:10.1080/03079458708436361,
PMid:18766600
Seal, B.S., King, D.J. & Bennett, J.D., 1995, ‘Characterization of Newcastle disease virus isolates by reverse transcription PCR coupled to direct
nucleotide sequencing and development of sequence database for pathotype prediction and molecular epidemiological analysis’, Journal of Clinical
Microbiology 33, 2624–2630.
PMid:8567895,
PMid:228544
Seal, B.S., King, D.J., Locke, D.P., Senne, D.A. & Jackwood, M.W., 1998, ‘Phylogenetic relationships among highly virulent Newcastle disease virus
isolates obtained from exotic birds and poultry from 1989 to 1996’, Journal of Clinical Microbiology 36, 1141–1145.
PMid:9542957,
PMid:104709
Sears, L.E., Moran, L.S., Kissinger, C., Creasey, T., Perry-O’Keefe, H., Roskey, M. et al., 1992, ‘CircumVent thermal cycle sequencing and
alternative manual and automated DNA sequencing protocols using the highly thermostable VentR (exo-) DNA polymerase’, Biotechniques 13, 626−633.
PMid:1476733
Shengquing, Y., Kishida, N., Ito, H., Kida, H., Otsuki, K., Kawaoka, Y. et al., 2002, ‘Generation of Velogenic Newcastle disease viruses from non-
pathogenic waterfowl isolates by passaging in chickens’, Virology 301, 206−211.
doi:10.1006/viro.2002.1539,,
PMid:12359423
Snoeck, C.J., Ducatez, M.F., Owoade, A.A., Faleke, O.O., Alakli, B.R., Tahita, M.C. et al., 2009, ‘Newcastle disease virus in West Africa: new virulent
strains identified in non-commercial farms’, Archives of Virology 154, 47−54.
doi:10.1007/s00705-008-0269-5,
PMid:19052688
Spradbrow, P.B., 1993, ‘Newcastle disease in village chickens’, Poultry Science Reviews 5, 57−96.
Spradbrow, P.B., 2000, ‘The Epidemiology of Newcastle Disease in Village Chickens’, in R.G. Alders & P.B. Spradbrow, SADC Planning
Workshop on Newcastle disease control in village chickens: Maputo, Mozambique, March 6–9, 2000, p. 53–55.
Tamura, K., Dudley, J., Nei, M. & Kumar, S., 2007, ‘MEGA4: molecular evolutionary Genetics analysis (MEGA) software version 4.0’, Molecular
Biology and Evolution 24, 1596−1599. doi:10.1093/molbev/msm092,
PMid:17488738
Takakuwa, H., Ito, T., Takada, A., Okazaki, K. & Kida, H., 1998, ‘Potentially virulent Newcastle disease viruses are maintained in migratory
waterfowl populations’, Japanese Journal of Veterinary Research 45, 207−215.
PMid:9553325
Thompson, J.D., Gibson, T.J., Plewniak, F., Jeanmougin, F. & Higging, D.G., 1997, ‘The CLUSTAL_X Windows interface: flexible strategies for multiple
sequence alignment aided by quality analysis tools’, Nucleic Acids Research 25, 4876−4882.
doi:10.1093/nar/25.24.4876,
PMid:9396791,
PMid:147148
Tollis, M. & Di Trani, L., 2002, ‘Review: Recent development in avian influenza research: Epidemiology and immunoprophylaxis’, Veterinary
Journal 164, 202−215.
doi:10.1053/tvjl.2002.0716,
PMid:12505393
Tsai, C., Pan, C., Liu, M., Lin, Y., Chen, C., Huang, T. et al., 2000, ‘Molecular epidemiological studies on Foot-and-Mouth disease type O Taiwan
viruses from 1997 epidemic’, Veterinary Microbiology 74, 207–216.
doi:10.1016/S0378-1135(00)00182-6
Van Regenmortel, M.H.V., Fauquet, C.M., Bishop, D.H.L., Carstens, E.B., Estes, M.K., Lemon, S.M. et al. (eds), 2000, ‘Virus Taxonomy.
Classification and Nomenclature of Viruses’, in 7th Report of the International Committee on Taxonomy of Viruses, International Union of
Microbiological Societies, Academic Press, London.
Wehmann, E., Czeglédi, A., Werner, O., Kaleta, E.F. & Lomniczi, B., 2003, ‘Occurrence of genotypes IV, V, VI and VIIa in Newcastle disease
outbreaks in Germany between 1939 and 1995‘, Avian Pathology 32, 157−163.
doi:10.1080/0307945021000071623,
PMid:12745369
Westbury, H., 2001, ‘Newcastle disease virus: an evolving pathogen?’, Avian Pathology 30, 5–11.
PMid:19184867
Yang, C.Y., Shieh, H.K., Lin, Y.L. & Chang, P.C., 1999, ‘Newcastle disease virus isolated from recent outbreaks in Taiwan phylogenetically related to
viruses (genotype VII) from recent outbreaks in Western Europe’, Avian Diseases 43, 125–130.
doi:10.2307/1592771,
PMid:10216769
Yongolo, M.G.S., 1996, ‘Epidemiology of Newcastle disease in village chickens in Tanzania’, MVM Dissertation, Department of Veterinary Microbiology and
Parasitology, Sokoine University of Agriculture.
Yongolo, M.G.S., Machangu, A.M. & Minga, U.M., 2002, ‘Newcastle disease and Infectious bursal disease among free range village chickens in Tanzania’,
in Characteristics and parameters of family poultry production in Africa., IAEA, Vienna p. 107−116, viewed 28 May 2011, from
http://www-naweb.iaea.org/nafa/aph/public/aph-poultry-africa.html
Yu, L., Wang, Z., Jinng, Y., Chang, L. & Kwang, J., 2001, ‘Characterisation of newly emerging Newcastle disease virus isolates from the People’s
republic of China and Taiwan’, Journal of Clinical Microbiology 39, 3512–3519.
doi:10.1128/JCM.39.10.3512-3519.2001,
PMid:11574565,
PMid:88381
|
|
