|
The aims of this study were to determine the modulating role of ascorbic acid (AA) on rectal temperature (RT), heterophil to lymphocyte (H to L)
ratio and aberrant behaviours of ostrich chicks transported by road for 4 h during hot-dry conditions. Twenty ostrich chicks aged 2.5 months, of
both sexes and belonging to the Red Neck breed, served as subjects of the study. The chicks were assigned randomly to AA-treated and control groups,
consisting of 10 chicks each. The AA-treated group was administered orally with 100 mg/kg body weight of AA dissolved in 5 mL of sterile water
30 min before transportation, whilst the control group was given the equivalent of sterile water only. The thermal load (TL) experienced in the
vehicle during transportation fluctuated between 31 °C and 89 °C, as calculated from the ambient temperature and relative humidity.
Transportation induced hyperthermia, lymphopenia, heterophilia and aberrant behaviours of pecking, wing fluffing and panting, which were ameliorated
by AA administration. The relationships between the TL, journey duration and physiological variables of RT, H to L ratio and aberrant behaviours
recorded during transportation were significantly and positively correlated in the control group. In AA-treated group the relationships were not
significantly correlated. In conclusion, the results showed for the first time that AA ameliorated the adverse effects of stress caused by road
transportation on the aberrant behaviours, RT and H to L ratio of ostrich chicks during the hot-dry season.
Transportation of animals is known to induce both psychological and physiological stress and stress-induced behavioural changes may result in
accompanying major physiological disturbances such as aberrant behaviours, immune-suppression, weight loss and disruption of general homeostasis
(Ayo et al. 2009; Minka & Ayo 2008, 2010; Stewart 1994). It has been established that changes in rectal temperature (RT), heterophil to
lymphocyte ratio (H to L) and behavioural activities of animals, including birds, are reliable indices of stress and are often used in the
development of management strategies aimed at increasing productivity. These changes are important for on-the-spot evaluation of the health status
and adaptability of animals to various stress factors, including transportation stress (Ayo, Oladele & Fayomi 2002; Minka & Ayo 2007a,
2007b, 2008; Warriss et al. 1993). Very often, behavioural changes are the main signs of distress and the first signs of disease in animals
(Ayo et al. 2002).The high levels of stress suffered by ostriches during road transportation has been sharply criticised by human rights and animal welfare activists
because this transportation poses a great risk to both the driver and the birds (Foggin 1992; Hoffman & Lambrechts 2011; Minka & Ayo 2008;
Wotton & Hewitt 1999). The lack of adequate recommendations relating solely to the welfare of ostriches during road transportation has
aggravated the problem (Ministry for Agriculture, Fisheries and Food 1998; Minka & Ayo 2008; Piccione et al. 2005). For example, in
Nigeria, ostriches are mostly purchased and transported by road from the Northern Guinea Savannah zone, where they are predominantly reared, to new
farms, markets, zoos and game reserves in other parts of the country (Ayo & Minka 2004; Minka 2003; Minka & Ayo 2007c, 2008). This road
transportation covers distances ranging from 200 km to over 1000 km and uses vehicles specifically not meant for transportation of ostriches, which
demonstrates little consideration for the ostriches’ welfare. In spite of the management procedures toward alleviation of transportation stress in ostriches, which centred solely on vehicle modification and
design, reports indicate that the birds still suffer considerable stress during transportation. In addition, the use of neuroleptics and other
central nervous depressants resulted in many ostriches developing rhabdomyopathy. Besides, the necessity of using such drugs in transported animals
is an indication that those animals’ welfare is already poor and compromised (Pfitzer & Lambrechts 2001). It has, therefore, become
necessary to search for safer agents that may alleviate the stresses ostriches suffer during their transportation by road without contravening their
welfare and productivity. Several studies have successfully employed ascorbic acid (AA), otherwise known as vitamin C, as an ameliorating agent against road transportation
stress in livestock, including poultry, because of its antioxidant and inhibitory properties on free radicals. The effects of stress are also
apparently reduced, as evidenced by lower cortisol levels (Ayo et al. 2009; Minka & Ayo 2007b, 2010, 2011). However, such studies in
ostriches are currently lacking in the available literature. Therefore, the aims of the present study were to evaluate the effects of a 4 h road
transportation on thermal load (TL), behaviour, RT and immunosuppression of ostrich chicks and to suggest AA as an ameliorating agent in ostrich
chicks transported by road during hot-dry conditions.
Thermal environmental and rectal
temperature data
The present study was conducted at the College of Agriculture and Animal Science, Kaduna, which is located at
11°10′ N, 07°38′ E in the Northern Guinea Savannah zone of Nigeria. The ambient temperature (AT), relative humidity (RH) and RT were recorded at the sites of the study for seven days before and after transportation,
at 07:00, 14:00 and 18:00 each day. These parameters were recorded hourly during the journey. The AT and RH were recorded using a wet-bulb and
dry-bulb thermometer (Thermometers Direct Ltd, Aldershot, United Kingdom). The RH was obtained by calculating the depression point from the
AT and wet-bulb temperature values. The temperature humidity index (THI), a measure of TL, recorded during the transportation period was obtained
from the AT and RH using a THI calculator. The RT was recorded with a digital clinical thermometer (Cocet, Kangfu Medical Equipment Co., Zhejiang,
China), inserted about 5 cm into the rectum via the cloaca for 3 min until an alarm was heard, indicating the end of the reading.
Experimental birds and measurements
Twenty apparently healthy, 2.5-month-old farmed ostrich chicks, belonging to the Red Neck breed and of both sexes, served as subjects. The birds
weighed 20.7 kg on average and were obtained from a standard commercial farm, located in Kaduna in the Northern Guinea Savannah zone of Nigeria.
The chicks were fed ostrich growers’ mash (Feed Master Ltd, Kaduna, Nigeria) with a nutritive value of 2465.0 kcal/kg metabolised energy,
22% protein and 12% fibre. They were given access to water ad libitum in both the farms from which and to which they were transported.
One week before transportation, the birds were assigned randomly into AA-treated and control groups, consisting of 10 chicks each. Blood samples
were obtained from the ostriches two days prior to transportation in order to obtain base-line (pre-transportation) values. On transportation day
and 30 min before loading, the AA-treated group was given 100.0 mg/kg body weight of AA orally, dissolved in 5 mL of sterile water, whilst control
group was given the equivalent of sterile water only. The 30 min time lapse from AA administration to loading was allowed for the absorption of the
AA before the commencement of the journey. The RT values of the ostriches were recorded 30 min before and after loading and subsequently every hour
of the transportation. During transportation, the behavioural events of individual chicks were recorded continuously, as described by Das,
Srivastava and Das (2001) and Minka and Ayo (2010). The behaviour was recorded by four observers inside the vehicle, with each observer recording
the on-going behaviour of five chicks in a similar pattern. The number of ostriches that exhibited behavioural events such as panting
(hyperventilation), wing fluffing and pecking were recorded whenever they were observed for each hour of the transportation period. Blood samples
were obtained immediately on arrival and on the third day after transportation. At each period of blood sampling 5.0 mL of blood was taken from a
wing vein into a heparinised test tube. Monolayer blood films were made from each sample, fixed and stained for differential leucocyte counts
(Dacie & Lewis 1975). Results for heterophils (H) and lymphocytes (L) are presented. The H to L ratio was calculated by dividing the value of H
by that of L.
Vehicle loading and transportation procedures
The ostriches were transported in a modified Bedford van. The floor of the van, which had no holes or gaps, was covered with chopped wood shavings,
on top of which was placed a non-slip rubber mat. The windows were covered with grills that permitted the regulation of ventilation. The van was
partitioned into 10 compartments with detachable and perforated plastic materials measuring 0.70 m high. Each compartment housed two ostrich chicks
comfortably and with enough space for them to lie down. Although recommendations on stocking density for ostrich chicks during transportation are
lacking (Wotton & Hewitt 1999), in the present study the chicks were stocked at a density of 0.35 m2 per chick. Each partition was
fixed in place after loading each ostrich into the vehicle. Similarly, the partitions were detached separately during unloading, starting from the
rear of the van and moving to the front. Transportation was undertaken during the hot-dry period of April. The vehicle travelled for 4 h – from 08:00 to 12:00 – covering a
distance of 220 km on an asphalt road, with an average speed of 50 km/h. Food and water were withdrawn completely 6 h before and during the journey
period. On arrival, the ostriches were immediately unloaded individually into a pen, where they were fed the same ration given to them before the
transportation.
Statistical analysis
The mean and standard error of different behavioural events were calculated. Data were subjected to the Student’s t-test, whilst
Pearson’s correlation analysis was employed in calculating the relationships between the journey periods, AT and behavioural activities.
Values of p < 0.05 were considered significant.
Ethical considerations
The handling, loading and transportation of the ostrich chicks were in accordance with national and international guidelines on humane handling and
transportation of animals and birds (Farm Animal Welfare Council 2003; Federal Republic of Nigeria 1988; MAFF 1998) and as described by Sales and
Smith (1995) and Wotton and Hewitt (1999). The College of Agriculture and Animal Science at the Ahmadu Bello University in Kaduna, Nigeria approved
all of the protocols required for the study period, under the permit number VCN-002519.
Potential benefits and hazards
Both physical and psychological trauma during transportation were anticipated because of the bi-pedal nature of the ostriches, which often leads to
traumatic injury, fracture and rabdomyolopathy. Thus, the birds were transported as recommended by the national and international guidelines cited
above. To ensure the safety of the birds, the primary author, a Veterinary Surgeon registered with the Veterinary Council of Nigeria, participated
fully in the transportation of the birds.
Thermal conditions and rectal temperature during transportation
The AT, RH and TL inside the vehicle rose with each hour of the journey, attaining their peak at the fourth hour when the transportation was
completed (Table 1). The RT recorded in AA-treated ostrich chicks 30 min after AA administration and just before loading rose (p < 0.05)
from the mean base-line value of 38.5 °C ± 0.2 °C to 39.6 °C ± 0.1 °C. Thereafter, the RT of the AA-treated birds
declined and fluctuated between the values of 38.5 °C and 39.0 °C during the post-loading and transportation periods and did not differ
(p > 0.05) from the base-line values. In the control group, the RT values recorded after loading and during each hour of the transportation
period fluctuated between 38.3 °C and 40.1 °C, significantly higher than the base-line values and corresponding values obtained in the
AA-treated ostriches (Figure 1a).
|
TABLE 1:
Fluctuations in thermal microenvironment variables inside the vehicle during a four-hour road transportation of ostriches.
|
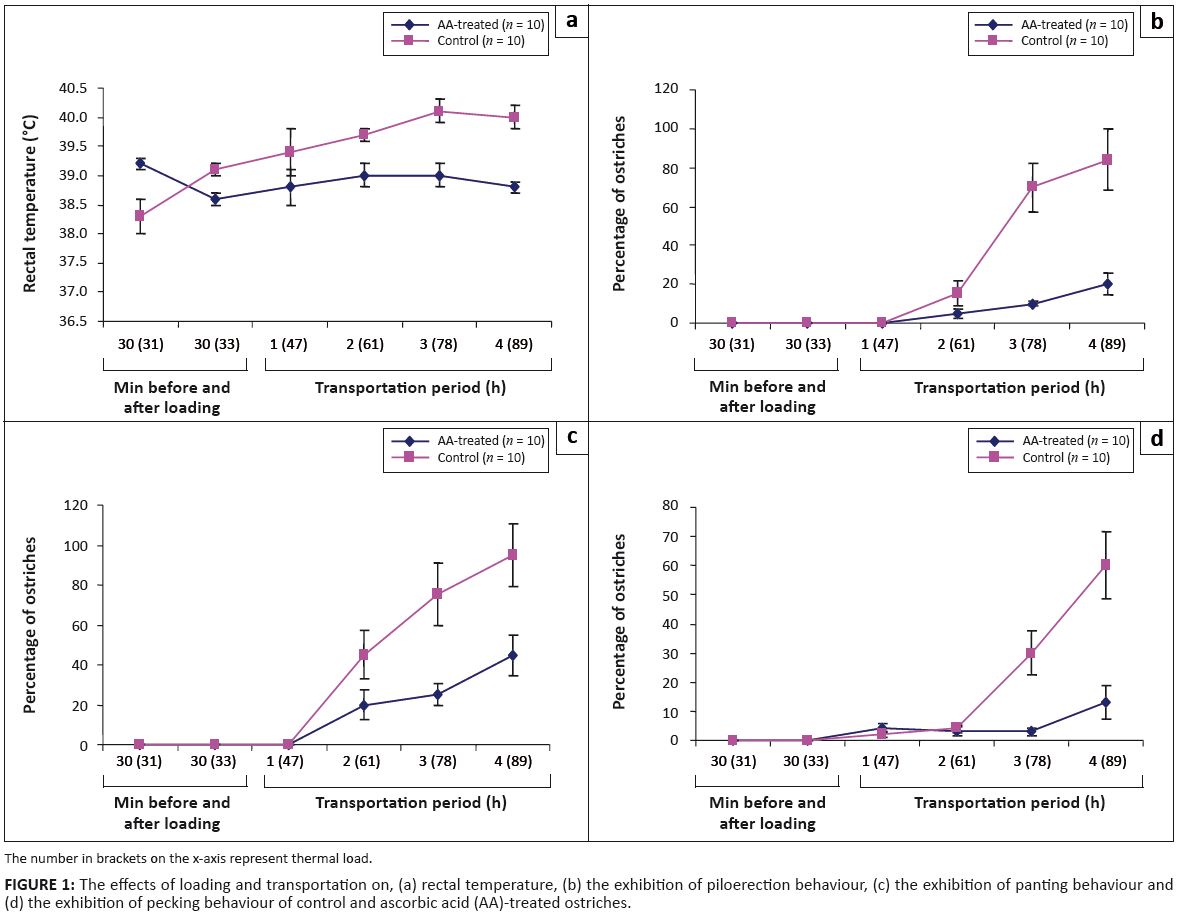 |
FIGURE 1:
The effects of loading and transportation on, (a) rectal temperature, (b) the exhibition of piloerection behaviour, (c) the exhibition of panting behaviour and
(d) the exhibition of pecking behaviour of control and ascorbic acid (AA)-treated ostriches.
|
|
Aberrant behaviours
The aberrant behaviours of piloerection (Figure 1b), panting (Figure 1c) and pecking (Figure 1d), recorded in the control ostriches increased
(p < 0.05) with the journey duration and TL; these behaviours manifested apparently during the last hours of the journey when the thermal
environment variables were at their peak. In AA-treated ostriches, panting was the only aberrant behaviour that significantly (p < 0.05)
manifested at the last hour of the journey.
Heterophil to lymphocyte ratio
The H to L ratio of 0.67 ± 0.30 recorded in the control chicks immediately after transportation was higher (p < 0.05) than the
base-line value of 0.33 ± 0.10 and the corresponding value of 0.34 ± 0.10 recorded in AA-treated chicks after transportation. The H
to L ratio recorded in the AA-treated group immediately and three days after transportation was not different from the base-line ratio. However,
the control chicks had still not returned to the base-line H to L ratio three days after transportation (Table 2).
|
TABLE 2:
Responses of heterophils and lymphocytes of ascorbic acid-treated (n = 10) and control (n = 10) ostriches to road transportation stress.
|
The relationships between the TL, journey duration and physiological variables of RT, H to L ratio and aberrant behaviour recorded during the
transportation were significantly and positively (p < 0.001; r = 0.814) correlated in the control group. In AA-treated group the
relationships were not significantly correlated (Table 3).
|
TABLE 3:
Relationships between the hour of the journey, thermal load, rectal temperature and aberrant behaviours in ascorbic acid-treated (n = 10)
and control (n = 10)
ostriches during the road transportation period.
|
The results obtained in the present study showed that the transportation of ostrich chicks during the hot-dry season is very stressful and poses a
risk to the health of the birds, findings which agreed with previous reports (Foggin 1992; Minka & Ayo 2007c, 2008; Mitchell et al. 1996).
The fact that the ostrich is known to withstand very high environmental temperatures without any consequential adverse effects on its homeostasis
(Stewart 1994) suggests that the mean AT value of 35 °C – recorded during transportation in the present study, and which was far below
the temperature (50 °C) at which ostriches begin to experience adverse effects to their homoeostasis – may not be a major cause of
transportation stress in the ostriches. This may be true because, unlike mammals and other birds, ostriches have excellent ability to thermoregulate
under high AT. However, during transportation, they suffer considerable stress when the AT values are either above or below 20 °C (Payne 1993;
Piccione et al. 2005), which is the recommended thermoneutral zone for ostriches (Skadhauge 1981).The mean TL value of 51.0 °C ± 7.8 °C and AT value of 33.6 °C ± 1.1 °C recorded during the journey were similar to
the TL and AT values of 44.0 °C and 34.0 °C, respectively, obtained during transportation of pullets, and which were reported to induce heat
stress in pullets (Minka & Ayo 2010). The fact that the TL increased with the hour of the day and journey duration suggests that as the journey
progressed the effect of TL on the birds also rose. In addition, panting behaviour of the birds increased the RH in the vehicle, which in turn
increased TL. This apparently exacerbated the mechanism of thermoregulation by increasing evaporative heat loss and inducing hyperthermia in the
ostriches. Other studies have shown that even at an AT of 18.0 °C and RH of 89% ostriches still suffer transportation stress (Payne 1993). It
appears therefore that other transportation factors such as handling, loading, vehicle vibration, motion, noise and novelty of environment may be
the major stress factors affecting the ostrich during transportation, rather than high AT and RH. This requires further investigation. The base-line values of RT recorded in both treated and control ostriches before transportation did not differ from each other and from the normal
range RT values of 38.3 °C to 39.3 °C established for ostrich chicks (Skadhauge 1981; Stewart 1994), although the RTs were at their lower
limit values. The results were similar to those obtained by Stewart (1994), who showed that ostriches, in contrast to other birds, have an RT below
40.0 °C and in the range of the eutherian mammals. The increase in the RT value recorded in AA-treated group 30 min after the administration of AA, but before loading, was within the normal range
for the ostrich, but was higher than that of the control, which demonstrated that administration of AA induced RT elevation in the ostriches. This
finding supports earlier results that AA exerts a biphasic effect on RT. Firstly, it increases the RT whenever the value is at a lower limit
threshold as observed in AA-treated ostriches in the present study. Secondly, it reduces RT values that are at the maximum threshold (Ayo, Minka
& Fayomi 2005; Ayo, Owoyele & Dzenda 2007). The increase in RT values recorded in the control group 30 min after handling and loading, but before the transportation, showed that these
procedures were stressful to the ostriches. Similar observations were made during the handling, loading and transportation of birds (Minka &
Ayo 2007b, 2011). The increase in RT value at this stage of transportation may not be associated with an increase in TL, but could be explained as
a result of physical activity; and also by stress-induced hyperthermia, which is the regulated shift of the thermoregulatory set-point mediated by
prostaglandin E and interleukin 1 and 6 (Kent & Ewbank 1983; Piccione et al. 2007). Stress-induced hyperthermia is associated with
cortisol release and is known to attain peak level within 10 min of administration of a stress factor (Piccione et al. 2007). The fact that a non-significant (p < 0.05) increase in RT value was recorded post-loading in the AA-treated ostriches suggested, for the
first time, that AA alleviated the effects of handling and loading stress on RT of the ostriches. The mechanism of action of AA on the handling and
loading stress factors may be caused by the fact that AA is an inhibitory vitaminergic neurotransmitter in the hypothalamus, which plays an
important role in thermoregulation by inhibiting cortisol, the chief hormone of stress (Balz 2003; Kannan et al. 2002). This is an important
finding because handling and loading of ostriches are the most critical procedures and the major cause of poor welfare (Minka & Ayo 2007c,
2008). The progressive increase in RT value during the transportation, especially in the control birds, suggested that transportation itself, apart from
the handling and loading procedures, further increased the release of cortisol, which apparently continued to alter RT. The present finding agrees
with those of Knowles et al. (1996), Hartung (2003) and Minka and Ayo (2008), who established that the period of transportation of an animal
is one of the most traumatic in its life. This is particularly true of ostriches, as they are very social birds and relatively minor changes in
their social status may cause severe stress (Stewart 1994). The effects of stress caused by road transportation acting concomitantly with high AT
and RH on the ostriches in the vehicle further aggravated the stressful conditions of the birds. The insignificant (p > 0.05) change in RT value recorded in the AA-treated ostriches, compared to those obtained in the control, further
indicated that AA did not only ameliorate the stress imposed by handling and loading, but also the stresses encountered during the actual process of
road transportation. Such stresses included vehicle motion, noise, vibration and speed, high AT and RH, long duration of journey, deprivation of
food and water, novelty of environment, and confinement. The findings of the present study demonstrated that AA reduced significantly the stress on
RT induced by road transportation. The results were similar to those obtained during transportation of ostriches and poultry by road (Minka &
Ayo 2008, 2010). Similar ameliorating effects of AA on the RT of pullets have been documented (Minka & Ayo 2010, 2011). The behavioural responses recorded during the third hour of transportation revealed some unusual and aberrant behaviours such as pecking (directed
to the ground or to objects), wing fluffing (piloerection) and panting (polypnoea), especially in the control ostriches. The manifestations of these
behaviours suggest that the ostriches were stressed and the level of stress increased with the duration of the journey and TL. Although ostriches
are known to have a wide range of thermoregulatory set points, it has been reported that ostrich chicks are affected severely by hot weather
(Minka & Ayo 2007c; Reiner, Seitz & Dzapo 1996; Stewart 1994), as confirmed by the present study. The aberrant behaviours of polypnoea and
piloerection were responses of the birds to increased demand in oxygen and heat dissipation brought about by the high AT and RH recorded in the
vehicle during the transportation. Because ostriches, like other birds, have no sweat glands, the only means available for cooling is evaporation
via polypnoea and piloerection by exposing the bare skin on their thorax and upper legs to enhanced convection and radiation losses. Similar
observations on the behaviours of ostriches exposed to different thermal conditions have also been reported (Samson 1996). Although the vehicle cabin was well ventilated, the air entering the vehicle was very hot and did not exert any cooling effect on the birds. This
is characteristic of the hot-dry season in the zone of study. The present result showed that ostriches began to pant when the TL value was
40 °C. The result obtained in AA-treated birds demonstrated that AA has reduced the effect of TL on the birds and facilitated the physiological
mechanism of thermoregulation. It has been established that AA decreases heat load through reduction in heat production or increasing heat loss by
enhancing thermal exchange between the body and the environment (Chang et al. 1993; Minka & Ayo 2010; Tauler et al. 2003).
Similarly, AA enhanced the adaptability of poultry and animals to environmental stress (McKee & Harrison 1995; Minka & Ayo 2010). The
mechanism of action of AA in reducing heat stress may also be through the detoxification of reactive oxygen species known to be in abundance in the
body during stressful conditions (Minka & Ayo 2007b; Tauler et al. 2003). The intense pecking behaviour exhibited by the ostriches in the control group may be associated with frustration, hunger and metabolic stress
caused by transportation procedures, which are tasking and completely novel to the birds (Foggin 1992). The fact that pecking behaviour was less
manifested in AA-treated birds suggested that AA reduced the bird’s frustration. AA is known to be the first vitaminergetic neurotransmitter
and it plays a significant role in inhibiting cortisol, the chief hormone of stress, and the limbic region involved in the elicitation of fear and
frustration (Balz 2003; Karanth et al. 2000). The significant (p < 0.05) increase in H to L ratio recorded in the control ostriches post-transportation indicated that the immune
system of the birds was compromised as a result of transportation up to the third day of the post-transportation period. The H to L ratio has been
used as the most common reliable index of stress measurement from blood analysis (Kannan et al. 2002; Minka & Ayo 2007b, 2011). A similar
effect of transportation stress on H to L ratio was reported in poultry, including ostriches (Minka & Ayo 2007b, 2008, 2011) and the increase in
H to L ratio has been reported to reflect the effects of elevated corticosteroids in the circulation induced by stress (Kannan et al. 2002;
Schalm, Jain & Carroll 1975). The insignificant change in H to L ratio obtained in AA-treated ostriches post-loading was an indication of
reduced physiological stress. The result showed, for the first time, that AA alleviated the stresses induced by both the transportation proper and
the post-transportation procedures on the lymphocytes and heterophils of the ostriches. AA has been reported to be a chain-breaking antioxidant,
involved in the prevention and restriction of free-radical chain formation and propagation. Consequently, AA enhances the immune status of the
ostriches by protecting the blood cells, including heterophils and lymphocytes, from oxidative damage induced by free radicals and cortisol
(Alok et al. 2003; Elsayed, Farghaly & Abu-Taleb 2008; Tauler et al. 2003). Although there are no recommended doses for AA as an
antioxidant in ostriches, it has been shown that it ameliorates heat stress in ostriches in supplementation, at a dose of 250 mg/kg of feed
(Elsayed et al. 2008).
The results of the present study have demonstrated for the first time that AA administration prior to the commencement of road transportation was
beneficial to ostrich chicks and may be used to improve the bird’s welfare and make their transportation more economically viable. For example,
AA ameliorated the adverse effects of stress caused by road transportation on the behaviour, RT and H to L ratio of ostrich chicks. It also
enhanced the homeostatic mechanisms of behavioural, thermoregulatory and cellular immune responses of ostrich chicks to the stressful conditions
imposed by road transportation during the hot-dry season. AA is readily available, cheap, safe and easy to administer in ostrich chicks, making it
an ideal supplement for use during transportation.
The authors are grateful to
all laboratory staff of the College of Agriculture and Animal Science at the Ahmadu Bello University, Kaduna for their technical assistance.
Competing interests
The authors declare that they have no financial or personal relationship(s) which may have inappropriately influenced them in writing this paper.
Authors’ contributions
M.N.S. (Ahmadu Bello University, Kaduna) was the project leader. He supervised the transportation, provided logistics, collected and prepared all
samples and data. He also provided an initial draft of the manuscript,
whilst A.J.O. (Ahmadu Bello University, Zaria) designed the project, statistically analysed the results and wrote the manuscript to standard.
Alok, K.B., Amrittal, M., Dipanjan, C. & Sajal, C., 2003, ‘Oxidants, antioxidants and physical exercise’, Molecular and
Cellular Biochemistry 253, 307–312. http://dx.doi.org/10.1023/A:1026032404105,
PMid:14619981
Ayo, J.O. & Minka, N.S., 2004, ‘Effect of six-hour road transportation on some physiological parameters in ostriches’,
Proceedings of the 29th Annual Conference of Nigerian Society for Animal Production, Sokoto, Nigeria, 21–25 March, pp.
58–61, Nigerian Society for Animal Production Publishers, Abuja. Ayo, J.O., Minka, N.S. & Fayomi, A., 2005, ‘Effects of ascorbic acid on rectal temperature of pullets transported by road during the
hot-dry season’, Tropical Journal of Animal Science 8, 43–48. Ayo, J.O., Minka, N.S., Sackey, A.K.B. & Adelaye, A.B., 2009, ‘Responses of serum electrolytes of goats to twelve hours of road
transportation during the hot-dry season in Nigeria, and the effect of pretreatment with ascorbic acid’, Onderstepoort Journal of
Veterinary Research 78, 409–418. Ayo, J.O., Oladele, S.B. & Fayomi, A., 2002, ‘Behavioural reactions of cattle to stress situations: A review’, Journal of
Agricultural Technology 8, 15–20. Ayo, J.O., Owoyele, O.O. & Dzenda, T., 2007, ‘Effects of ascorbic acid on diurnal variations in rectal temperature of Brown
Nera pullets during the harmattan season’, International Journal of Poultry Science 6, 612–616.
http://dx.doi.org/10.3923/ijps.2007.612.616 Balz, F., 2003, ‘Vitamin C intake’, Nutritional Disease 14, 1–18. Chang, K.C., Chong, W.S., Sohn, D., Kwon, B.H., Lee, I.J., Kim, C.Y. et al., 1993, ‘Endothelial potentiation of relaxation response
to ascorbic acid in rat and guinea pig thoracic aorta’, Life Science 52, 37–42. Dacie, J.U. & Lewis, S.M., 1975, Practical haematology, 5th edn., Churchill Livingstone, Edinburgh. Das, K.S., Srivastava, B.B. & Das, N., 2001, ‘Standing orientation and behaviour of goats during short-haul road transportation’,
Small Ruminant Research 41(1), 91–94.
http://dx.doi.org/10.1016/S0921-4488%2801%2900187-0
Elsayed, M.A., Farghaly, H.A.M. & Abu-Taleb, A.M., 2008, ‘Effect of vitamin C supplementation on some physiological parameters of
ostrich exposed to early heat stress of temperature’, Applied Life Science 40(4S2), 1503–1513. Farm Animal Welfare Council, 2003, Farm animal at slaughter or killing: Part 1, Defra Publications, London. Federal Republic of Nigeria, 1988, Animal Disease (Control) Decree (Act No. 10 of 1988), Government Gazette Feb, A477–501,
Government Printers, Lagos. Foggin, C.M., 1992, ‘Veterinary problems of ostriches’, in M.G. Hallam (ed.), The Topaz introduction to practical ostrich
farming, pp. 61–96, Superior Print, Harare. Hartung, J., 2003, ‘Effects of transport on health of farm animals’, Veterinary Research Communication 27, 525–527.
http://dx.doi.org/10.1023 B:VERC.0000014212.81294.78,
PMid:14535462
Animal Welfare 11, 195–235.
http://dx.doi.org/10.1007/978-3-642-19297-5_10
Kannan, G., Terril, T.H., Kouokou, B., Gelaye, S. & Amoah, E.A., 2002, ‘Simulated preslaughter holding and isolation effects on stress
responses and liveweight shrinkage in meat goats’, Journal of Animal Science 80, 1771–1780.
PMid:12162644
Karanth, S., Yu, W.H., Walczewska, A., Mastronardi, C. & McCann, SM., 2000, ‘Ascorbic acid acts as an inhibitory transmitter in the
hypothalamus to inhibit stimulated lutenizing hormone-releasing hormone release by scavenging nitric oxide’, Proceedings of
the Academy of Sciences of the United State of America 97, 1891–1896. Kent, J.E. & Ewbank, R., 1983, ‘The effect of road transportation on the constituent and behaviour of calves: 1. Six months old’,
British Veterinary Journal 139, 228–235. Knowles, T.G., Ball R.C., Warriss, P.D. & Edwards, J.E., 1996, ‘A survey to investigate potential dehydration in slaughtered broiler
chickens’, British Veterinary Journal 152, 307–314.
http://dx.doi.org/10.1016/S0007-1935(96)80102-3 Ministry for Agriculture, Fisheries and Food, 1998, Guidance on the welfare of animals (transport) Order 1997, No. PB 3766, Her
Majesty’s Stationery Office, London. McKee, J.S. & Harrison, P., 1995, ‘Effects of supplemental ascorbic acid on the performance of broilers chickens exposed to multiple
stressors’, Poultry Science 74, 1772–1785.
PMid:8614686 Minka, N.S., 2003, ‘Evaluation of the performance of ostrich chicks in northern Nigeria’, Tropical Journal of Animal Science
6, 69–73. Minka, N.S. & Ayo, J.O., 2007a, ‘Effects of loading behaviour and road transport stress on traumatic injuries in cattle transported by
road during the hot-dry season’, Livestock Science 107, 91–95.
http://dx.doi.org/10.1016/j.livsci.2006.10.013 Minka, N.S. & Ayo, J.O., 2007b, ‘Haematology and behaviour of pullets transported by road and administered with ascorbic acid during the
hot-dry season’, Research in Veterinary Science 85(2), 389–393.
http://dx.doi.org/10.1016/j.rvsc.2007.11.009,
PMid:18179807
Minka, N.S. & Ayo, J.O., 2007c, ‘Road transportation effect on rectal temperature, respiration and heart rates of ostrich (Struthio
camelus) chicks’, Veterinarski Arhiv 77, 39–46. Minka, N.S. & Ayo, J.O., 2008, ‘Assessment of the stresses imposed on adult ostriches (Struthio camelus) during handling,
loading, transportation and unloading’, Veterinary Record 162, 846–851.
http://dx.doi.org/10.1136/vr.162.26.846,
PMid:18587061
Minka, N.S. & Ayo, J.O., 2010, ‘Behavioural and rectal temperature responses of Black Harco pullets administered vitamin C and E and
transported by road during the hot-dry season’, Journal of Veterinary Behavior and Clinical Applications and Research 5, 134–144.
http://dx.doi.org/10.1016/j.jveb.2009.12.022 Minka, N.S. & Ayo, J.O., 2011, ‘Modulating role of vitamins C and E against transport-induce stress in pullets during the hot-dry
conditions’, ISRN Veterinary Science 2011, Art. #497138, 7 pages.
http://dx.doi.org/10.5402/2011/497138 Mitchell, M.A., Kettlewell, P.J., Sandercock, R. & Spackman, D.A., 1996, ‘Physiological stress in ostriches during road
transportation’, in D.C. Deeming (ed.), Improving our understanding of ratites in farming environment, Ratite Conference
proceedings, 27–29 March, University of Manchester, UK, pp. 79–80. Payne, H., 1993, ‘Welfare of ostriches and other ratites during transport’, State Veterinary Journal 3(4), 8–12. Pfitzer, S. & Lambrechts, H., 2001, ‘The use of haloperidol during transport of adult ostriches’, Journal of the South African
Veterinary Association 72(1), 2–3.
PMid:11563712
Piccione, G., Coste, A., Gurlice, E. & Caola, G., 2005, ‘Preliminary investigation into thermal stress during diurnal road transportation
of young ostriches’, Archiv für Tierzucht 48, 194–200. Piccione, G., Fazio, F., Giannett, C., Assenza, A. & Caola, G., 2007, ‘Oxidative stress in thoroughbreds during official 1800-metre
races’, Veterinarski Arhiv 77(3), 219–227. Reiner, G., Seitz, K. & Dzapo, V., 1996, ‘A survey of farming environment and ostrich behaviour’, in D.C. Deeming (ed.),
Improving our understanding of ratites in farming environment, Ratite Conference proceedings, 27–29 March, University of
Manchester, UK, pp. 52–56. Sales, J. & Smith, W.A., 1995, Incubation and management of ostrich and ostrich products, University Printers, Stellenbosch. Samson, J., 1996, ‘Behavioural problems in farmed ostriches in Canada’, Canadian Veterinary Journal 37, 412–414.
PMid:8809393, PMCid:1576451 Schalm, O.W., Jain, N.C. & Carroll, E.J., 1975, Veterinary haematology, 3rd edn., Lea and Febiger, Philadelphia. Skadhauge, E., 1981, Osmoregulation in birds, Springer-Verlag, Berlin. Stewart, J.S., 1994, ‘Ostrich behaviour and behavioural problems’, Proceedings of the Annual Conference of Avian
Veterinarians, 28–30 September, Reno, USA, pp. 104–109, Association of Avian Veterinarians, Denver. Tauler, P., Aguilo, A., Gimeno, I., Fuentespina, E., Tur, J.A. & Pons, A., 2003, ‘Influence of vitamin C diet supplementation on
endogenous antioxidant defense during exhaustive exercise’, European Journal of Physiology 446, 658–664. Warriss, P.D., Kestin, S.C., Brown, S.N., Knowles, T.G., Wilkins, L.J., Edwards, J.E. et al., 1993, ‘The depletion of glycogen
stress and indices of dehydration in transported broilers’, Veterinary Record 149, 391–398. Wotton, S.B. & Hewitt, L., 1999, ‘Transportaton of ostriches – A review’, Veterinary Record 145, 725–731.
PMid:10972110
|
